ORIGINAL RESEARCH
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Box. Major Content Areas of the Arkansas Cardiovascular Health Examination Survey Questionnaires and Physical Examination |
|---|
Questionnaire Domains
Physical Examination
Blood and Urine Tests
|
The ARCHES questionnaire consisted of up to 285 questions (depending on skip patterns) covering behavioral, psychosocial, socioeconomic, and demographic variables, personal and family medical history, cardiovascular and other chronic disease risk factors, health care access, and other subjects (Box). Questions were mainly from BRFSS (5) and NHANES (6). The complete questionnaire was pretested through cognitive interviews with a convenience sample of low-income community volunteers, and necessary changes were made. Nutrient intake data were collected by using the FFQ developed by the Nutrition Assessment Shared Resource (7) of the Fred Hutchinson Cancer Research Center (FHCRC). Questionnaires are available on the ARCHES page of the ADH website (http://www.healthy.arkansas.gov/ programsServices/chronicDisease/Initiatives/ Pages/Arches.aspx).
After the participant provided written informed consent, interviewers administered the questionnaire and recorded responses on paper forms. Interviewers also examined medicine bottles for all medications (prescribed and over-the-counter, including dietary supplements), and recorded medication names. We measured participants’ height, weight, and abdominal circumference by using standard NHANES protocols, while they wore light clothing and no shoes. We used a Tanita digital, self-calibrating scale (model HD-351) (Tanita Corporation of America, Inc, Arlington Heights, Illinois) to measure weight. After measurement of arm circumference and use of an appropriate-sized cuff, interviewers recorded blood pressure 3 times at intervals during the interview process by using standard protocols (6), with an Omron HEM-907XL monitor (Omron Healthcare, Inc, Bannockburn, Illinois). For each recording, the monitor recorded the average of 3 separate readings, for up to 9 readings and 3 recordings.
After administering the questionnaire and taking anthropometric measurements, interviewers collected blood and urine samples. Interviewers placed the samples in containers with frozen-gel bags, processed them in the field according to protocols provided by Clinical Reference Laboratories, Inc (CRL) (Lenexa, Kansas), and shipped them to CRL, a laboratory certified according to the CDC Lipid Standardization Program (http://www.cdc.gov/labstandards/lsp.html), for analysis and reporting. The laboratory also froze aliquots of blood and urine and shipped them to ADH for storage and future analyses.
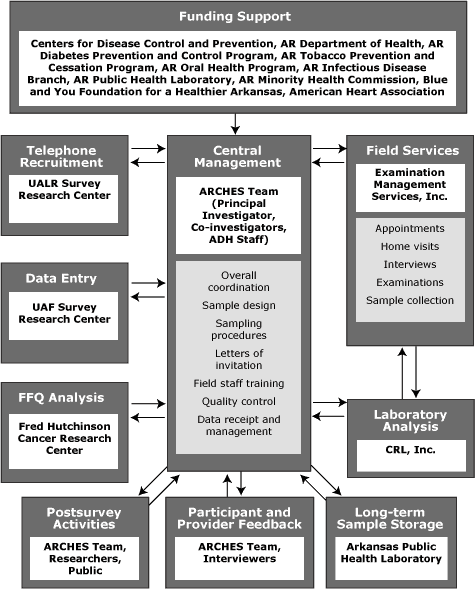
We managed all activities, including tracking of interviews, movement of forms and biological samples, data entry, and quality control, centrally at ADH, through contracts with several external entities (Figure 1). After completing the interview, EMSI personnel shipped questionnaires, forms, and biological samples to CRL, where blood and urine samples were further processed and analyzed. CRL sent the results of analyses, along with forms and questionnaires, to ADH, where they were logged and entered in the main database. The University of Arkansas at Fayetteville Survey Research Center scanned the main questionnaires, digitized the data, and transmitted them electronically to ADH. ADH personnel logged and shipped the FFQs that had been mailed back by participants to FHCRC for analysis. FFQ results were electronically transmitted back to ADH. After all data were received and entered into the main database, we created an analysis dataset, stripped of personal identifiers, for use in further analyses.
We took several steps to ensure data integrity and quality. One of the senior investigators telephoned a 5% random subsample of respondents within a few days of the interviews to ascertain interviewer accuracy and find out about participants’ experiences with the interviewers. We reported inaccuracies or problems to EMSI for corrective action. Before scanning and data entry, we hand-checked all questionnaires for readability, correct skip patterns, and missing data, and made corrections to the extent possible (calling respondents when needed). We pulled a 5% random subsample of paper copies of the main questionnaires and checked them against the electronic data to ensure accuracy in digitization. We hand-checked all FFQs before scanning and analysis. Laboratory analyses were subject to internal laboratory standards and checks by CRL and to range and consistency checks of all data by ARCHES staff.
Interviewers informed respondents with high blood pressure of their measurements and advised them to seek appropriate medical care. We monitored the results of blood and urine tests as they were received by ARCHES; respondents with results above predetermined critical values were contacted immediately and advised to seek medical attention, with an offer to fax results directly to their health care provider. We sent all blood and urine results, within 2 weeks of receipt, to each respondent along with a cover letter, with abnormal values flagged for attention and discussion with their health care provider.
Results
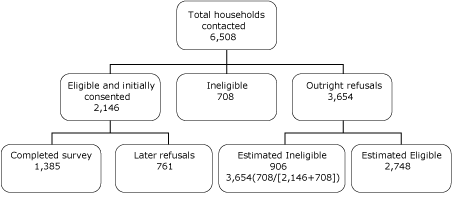
Of the 6,508 households contacted by telephone, 4,894 were eligible. Of these, refusals accounted for 2,748, and 2,146 gave initial consent to participate, for an initial response rate of 44%. The final number of completed household visits was 1,385, resulting in a CASRO (Council of American Survey Research Organizations) response rate of 28.3% (Figure 2).
Figure 2. Response rate calculation (using Council of American Survey Research Organizations formula) for Arkansas Cardiovascular Health Examination Survey sample. The response rate was the number who completed surveys (1,385) divided by the sum of the number who were eligible and initially consented (2,146) plus the estimated number who were eligible among those who refused (2,748): 1,385/(2,146 + 2,748) = 28.3%. [A text description of this figure is also available.]
Among completed visits, 1,265 (91.3%) participants also mailed in their FFQs, and 1,202 (86.8%) gave consent to freezing biological samples for future analyses. Also, 1,115 (80.5%) consented to future contact for follow-up surveys.
Compared with the state population, the ARCHES sample had a higher proportion of women (66.9% vs 51.7%), a higher proportion of blacks (23.8% vs 14.9%), and an older age distribution (Table 2). Median annual household income for the sample, about $35,000, was just slightly lower than the state median income of about $36,600 in 2007 (8), the time when the sample was developed.
Discussion
In organizing ARCHES, we had 3 goals: 1) to foster collaboration among programs, 2) to leverage CDC funds, and 3) to minimize the burden on resources of the health department. We achieved the first 2 goals by approaching a number of internal programs and external partners and encouraging their participation. This resulted in additional cash funding from 8 entities (Figure 1), with a 1.42:1 ratio of total cash available to that provided by CDC. We achieved the third goal by contracting with different entities to carry out much of the work, such as initial recruitment, setting appointments, conducting interviews and examinations, data entry, and laboratory analyses (Figure 1). Using this approach, each completed survey cost about $780. However, as a potential future ongoing surveillance effort, efficiency can be improved by using a different approach, focusing on higher priority data collection, and eliminating sample storage.
The tasks of overall coordination, data management, and quality control were maintained by the staff at ADH, directed by the principal investigator and the co-investigators ? a collaboration between ADH, the Fay W. Boozman College of Public Health at the University of Arkansas for Medical Sciences, and the Arkansas Minority Health Commission. This aspect of ARCHES, the ability to partner with many entities, was a major accomplishment of the project and a factor in its successful completion.
We completed interviews and examinations on 1,385 people, exceeding the goal of 1,344. Our sample was older and had a higher proportion of women than the Arkansas adult population (9). This is not surprising for a household survey (10) and was probably further influenced by our use of landline telephones only. At the time of sampling, cellular telephone numbers linked to addresses at census tract level (our primary sampling unit) were not publicly available; therefore, we were compelled to use landlines. Also, our BRFSS survey in 2007 was landline-based, facilitating eventual comparison of ARCHES results with BRFSS. The higher proportion of blacks in ARCHES was by design, fulfilling the CDC requirement of oversampling 1 priority population. All 3 of these differences were taken into account in the individual survey sampling weights, and the use of analytic software that allows accommodation for these sampling factors. Approximately one-third of those giving initial consent were either unable to be scheduled or declined to be scheduled for the in-home visit. This rate is close to that of the national REGARDS study, which used similar methods (11). Although the 28% CASRO response rate may be of some concern, there are no similar published state-level studies for comparison. However, the acceptable range of individual sampling weights (0.23-3.08) and the similar household incomes of participants and nonparticipants increase our confidence in the representativeness of the weighted sample.
ARCHES has provided ADH with a large amount of data that are being used to generate much-needed health information. Some of this information, such as levels of undiagnosed or uncontrolled disease (eg, hypertension, diabetes) and risk factors (such as overweight and obesity), is available for the first time at the state level. The data differ greatly from those obtained from self-reported BRFSS surveys. For example, obesity, hypertension, and diabetes are 50% to 55% more prevalent than indicated by BRFSS data (unpublished data). The data provide the ADH programs, researchers, and clinicians with information necessary to address the state’s worsening public health problems. Already the data have been used to inform policy makers and legislators about the more prevalent chronic diseases in the state and have resulted in a legislative request for an interim study of hypertension in Arkansas. The data are also being used by ADH, public health researchers, and students to analyze specific patterns of consumption, risk factors, and diseases in the state, leading to program development and improvement. In addition, the bank of frozen biological specimens will be used for future studies of risk factors and diseases in the state.
Conducting a state-level health examination survey was challenging. Arkansas’s model shows that by working with internal and external partners, and by contracting out major survey tasks, it is possible to conduct such a health examination survey without putting undue burden on the human and financial resources of the health department. The local information provided can be of benefit in addressing chronic diseases at the state level. This benefit, however, can be fully realized only if such surveys are repeated regularly, allowing states to track changes and effects of policies and programs. On the basis of our experience with ARCHES, we believe that repeating such surveys approximately every 5 years is feasible and has the potential to provide timely information for monitoring progress toward intermediate and long-term goals related to outcomes.
Acknowledgments
We thank the staff at the following institutions for their help in various aspects of the survey: ADH, University of Arkansas for Medical Sciences Fay W. Boozman College of Public Health, Arkansas Minority Health Commission, Abbott Renal Care Inc, EMSI, CRL, the Survey Research Centers of the University of Arkansas at Little Rock and at Fayetteville, Fred Hutchinson Cancer Research Center, American Heart Association, and Blue and You Foundation for a Healthier Arkansas. Direct funding support for ARCHES was received from CDC, Abbott Renal Care Inc, Blue and You Foundation for a Healthier Arkansas, Arkansas Minority Health Commission, and the following programs at the Arkansas Department of Health: Tobacco Prevention and Cessation Program, Diabetes Prevention and Control Program, and the Oral Health Program.
Author Information
Corresponding Author: Namvar Zohoori, MD, MPH, PhD, Chronic Disease Director and Associate Director of Science, Center for Health Advancement, Arkansas Department of Health, 4815 West Markham St, Slot 6, Little Rock, AR 72205. Telephone: 501-661-2546. E-mail: Namvar.Zohoori@arkansas.gov.
Author Affiliations: LeaVonne Pulley, Fay W. Boozman College of Public Health, University of Arkansas for Medical Sciences, Little Rock, Arkansas; Camille Jones, Arkansas Minority Health Commission, Little Rock, Arkansas; John Senner, Arkansas Department of Health, Little Rock, Arkansas; Hylan Shoob, Robert K. Merritt, Centers for Disease Control and Prevention, Atlanta, Georgia.
References
- Thorpe LE, Gwynn RC, Mandel-Ricci J, Roberts S, Tsoi B, Berman L, et al. Study design and participation rates of the New York City Health and Nutrition Examination Survey, 2004. Prev Chronic Dis 2006;3(3). http://www.cdc.gov/pcd/issues/2006/jul/05_0177.htm. Accessed April 22, 2010.
- Kish L, Anderson DW. Multivariate and multipurpose stratification. J Am Stat Assoc 1978;73(361):24-34.
- Kish L. A procedure for objective respondent selection within the household. J Am Stat Assoc 1949;44(247):380-7.
- National Center for Health Statistics. Vintage 2007 bridged-race postcensal population estimates. http://www.cdc.gov/nchs/nvss/bridged_race/data_documentation.htm#vintage2007. Accessed January 15, 2010.
- Behavioral Risk Factor Surveillance System survey questionnaire. Atlanta (GA): US Department of Health and Human Services, Centers for Disease Control and Prevention; 2006.
- National Health and Nutrition Examination Survey questionnaire and examination protocol. Atlanta (GA): US Department of Health and Human Services, Centers for Disease Control and Prevention; 2005-2006. http://www.cdc.gov/nchs/nhanes/nhanes2005-2006/nhanes05_06.htm. Accessed November 12, 2005.
- Schakel SF. Maintaining a nutrient database in a changing marketplace: keeping pace with changing food products — a research perspective. J Food Comp Anal 2001;14:315-22.
- Small area income and poverty estimates. Final release date 2008. Washington (DC): US Census Bureau. http://quickfacts.census.gov/qfd/states/05000lk.html. Accessed May 2010.
- Blumberg SJ, Luke JV. Wireless substitution: early release of estimates from the National Health Interview Survey, July-December 2008. National Center for Health Statistics. http://www.cdc.gov/nchs/data/nhis/earlyrelease/wireless200905.htm. Accessed April 21, 2010.
- Centers for Disease Control and Prevention. Behavioral Risk Factor Surveillance System 2009 summary data quality report (version #1 – Revised 04/27/2010). ftp://ftp.cdc.gov/pub/Data/Brfss/2009_Summary_Data_Quality_Report.pdf. Accessed April 22, 2010.
- Howard VJ, McClure LA, Meschia JF, Pulley L, Orr SC, Friday GH. High prevalence of stroke symptoms among persons without a diagnosis of stroke or transient ischemic attack in a general population: the REasons for Geographic And Racial Differences in Stroke (REGARDS) study. Arch Intern Med 2006;166(18):1952-8.
Tables
 Table 1. Timeline of Contact With Participants and Main Activities of the Arkansas Cardiovascular Health Examination Survey (ARCHES)
Table 1. Timeline of Contact With Participants and Main Activities of the Arkansas Cardiovascular Health Examination Survey (ARCHES)
| Stage of Survey | Mode of Contact With Participant | Activities Conducted (Dates Accomplished) | Organization and Personnel Involved |
|---|---|---|---|
| Sampling stage 1 | NA | Selected 375 of 623 census tracts and oversampled tracts with highest proportions of blacks. (May 2006) | ADH, ARCHES staff, and statistician |
| Sampling stage 2 | Letters to random sample of households within selected clusters | Informed about the survey, included informational brochure, and told to expect telephone call. (June 2006-October 2007) | ADH, ARCHES staff |
| Sampling stage 3 | Initial telephone calls to households in random order | Provided additional information and answered questions, obtained initial household consent, selected participating adult, obtained participant’s initial verbal consent to participate; maximum 4 per cluster. (June 2006-February 2008) | University of Arkansas at Little Rock, Survey Research Center |
| Field work | Calls to participants to make appointment for interview | Made appointment for home visit, provided additional information about the interview and exam, and gave instructions about fasting for blood draw. (July 2006-March 2008) | Examination Management Services, Inc, call center |
| Home visits | Obtained written informed consent, completed questionnaire, performed anthropometric and blood pressure measurements, and collected blood and urine samples. (July 2006-March 2008) | Examination Management Services, Inc, interviewers | |
| Laboratory analysis and data entry | NA | Analyzed blood and urine samples and transmitted electronically to ADH. (July 2006-April 2008) | Clinical Reference Laboratories, Inc |
| Scanned and digitized main questionnaire data and transmitted electronically to ADH. (July 2006-July 2008) | University of Arkansas at Fayetteville, Survey Research Center | ||
| Scanned and analyzed food frequency questionnaire data and transmitted electronically to ADH. (July 2006-July 2008) | Fred Hutchinson Cancer Research Center | ||
| Telephone calls to some participants | Verified data for quality control purposes and to complete missing or discrepant data. (July 2006-March 2008) | ADH, ARCHES staff | |
| Reporting to participants | Telephone calls to some participants | Called participants with critical values within 24 hours of receipt of results. (July 2006-March 2008) | ADH, principal investigator |
| Letters to all participants | Included copy of all blood and urine results, thank-you letter and general explanation of results, with instructions for further follow-up with primary care provider. Also included gift cards. (July 2006-May 2008) | ADH, ARCHES staff |
Abbreviation: NA, not applicable; ADH, Arkansas Department of Health.
 Table 2. Demographic Characteristics of the Arkansas Cardiovascular Health Examination Survey (ARCHES) Sample and of the Arkansas Adult Population, 2007
Table 2. Demographic Characteristics of the Arkansas Cardiovascular Health Examination Survey (ARCHES) Sample and of the Arkansas Adult Population, 2007
| Characteristic | No. of ARCHES Respondents (%) | 2007 Arkansas Adult Population, %a |
|---|---|---|
| Sex | ||
| Men | 459 (33.1) | 48.3 |
| Women | 926 (66.9) | 51.7 |
| Race | ||
| White | 1,056 (76.2) | 85.1 |
| Black | 329 (23.8) | 14.9 |
| Age, y | ||
| 18-49 | 430 (31.0) | 57.0 |
| 50-64 | 540 (39.0) | 24.2 |
| =65 | 415 (30.0) | 18.8 |
a Source: National Center for Health Statistics (4).
Source URL: http://www.cdc.gov/pcd/issues/2011/may/10_0134.htm
Source Agency: Centers for Disease Control and Prevention (CDC)
Captured Date: 2016-05-23 21:56:28.0