
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Summary of Notifiable Diseases --- United States, 2007
Please note: An erratum has been published for this article. To view the erratum, please click here. Prepared by Patsy A. Hall-Baker, Coordinator, Summary of Notifiable Diseases1 Enrique Nieves, Jr., MS1 Ruth Ann Jajosky, DMD1 Deborah A. Adams1 Pearl Sharp1 Willie J. Anderson1 J. Javier Aponte1 Gerald F. Jones1 Aaron E. Aranas, MPH, MBA1 Araceli Rey, MPH1 Bernetta Lane, MBA1 Michael S. Wodajo2 1Division of Integrated Surveillance Systems and Services, National Center for Public Health Informatics, Coordinating Center for Health Information and Service, CDC 2McKing Consulting Corporation PrefaceThe Summary of Notifiable Diseases --- United States, 2007 contains the official statistics, in tabular and graphic form, for the reported occurrence of nationally notifiable infectious diseases in the United States for 2007. Unless otherwise noted, the data are final totals for 2007 reported as of June 30, 2008. These statistics are collected and compiled from reports sent by state health departments and territories to the National Notifiable Diseases Surveillance System (NNDSS), which is operated by CDC in collaboration with the Council of State and Territorial Epidemiologists (CSTE). The Summary is available at http://www.cdc.gov/mmwr/summary.html. This site also includes publications from previous years. The Highlights section presents noteworthy epidemiologic and prevention information for 2007 for selected diseases and additional information to aid in the interpretation of surveillance and disease-trend data. Part 1 contains tables showing incidence data for the nationally notifiable infectious diseases during 2007.* The tables provide the number of cases reported to CDC for 2007 and the distribution of cases by month, geographic location, and the patient's demographic characteristics (age, sex, race, and ethnicity). Part 2 contains graphs and maps that depict summary data for certain notifiable infectious diseases described in tabular form in Part 1. Part 3 contains tables that list the number of cases of notifiable diseases reported to CDC since 1976. This section also includes a table enumerating deaths associated with specified notifiable diseases reported to CDC's National Center for Health Statistics (NCHS) during 2002--2005. The Selected Reading section presents general and disease-specific references for notifiable infectious diseases. These references provide additional information on surveillance and epidemiologic concerns, diagnostic concerns, and disease-control activities. Comments and suggestions from readers are welcome. To increase the usefulness of future editions, comments about the current report and descriptions of how information is or could be used are invited. Comments should be sent to Public Health Surveillance Team --- NNDSS, Division of Integrated Surveillance Systems and Services, National Center for Public Health Informatics at soib@cdc.gov. BackgroundThe infectious diseases designated as notifiable at the national level during 2007 are listed in this section. A notifiable disease is one for which regular, frequent, and timely information regarding individual cases is considered necessary for the prevention and control of the disease. A brief history of the reporting of nationally notifiable infectious diseases in the United States is available at http://www.cdc.gov/ncphi/disss/nndss/nndsshis.htm. In 1961, CDC assumed responsibility for the collection and publication of data on nationally notifiable diseases. NNDSS is neither a single surveillance system nor a method of reporting. Certain NNDSS data are reported to CDC through separate surveillance information systems and through different reporting mechanisms; however, these data are aggregated and compiled for publication purposes. Notifiable disease reporting at the local level protects the public's health by ensuring the proper identification and follow-up of cases. Public health workers ensure that persons who are already ill receive appropriate treatment; trace contacts who need vaccines, treatment, quarantine, or education; investigate and halt outbreaks; eliminate environmental hazards; and close premises where spread has occurred. Surveillance of notifiable conditions helps public health authorities to monitor the impact of notifiable conditions, measure disease trends, assess the effectiveness of control and prevention measures, identify populations or geographic areas at high risk, allocate resources appropriately, formulate prevention strategies, and develop public health policies. Monitoring surveillance data enables public health authorities to detect sudden changes in disease occurrence and distribution, identify changes in agents and host factors, and detect changes in health-care practices. The list of nationally notifiable infectious diseases is revised periodically. A disease might be added to the list as a new pathogen emerges, or a disease might be deleted as its incidence declines. Public health officials at state health departments and CDC collaborate in determining which diseases should be nationally notifiable. CSTE, with input from CDC, makes recommendations annually for additions and deletions. Although disease reporting is mandated by legislation or regulation at the state and local levels, state reporting to CDC is voluntary. Reporting completeness of notifiable diseases is highly variable and related to the condition or disease being reported (1). The list of diseases considered notifiable varies by state and year. Current and historic national public health surveillance case definitions used for classifying and enumerating cases consistently across reporting jurisdictions are available at http://www.cdc.gov/ncphi/disss/nndss/nndsshis.htm. Infectious Diseases Designated as Notifiable at the National Level During 2007*Acquired immunodeficiency syndrome (AIDS) Anthrax Domestic arboviral diseases California serogroup virus disease Eastern equine encephalitis virus disease Powassan virus disease St. Louis encephalitis virus disease West Nile virus disease Western equine encephalitis virus disease Botulism foodborne infant other (wound and unspecified) Brucellosis Chancroid Chlamydia trachomatis, genital infection Cholera Coccidioidomycosis Cryptosporidiosis Cyclosporiasis Diphtheria Ehrlichiosis human granulocytic human monocytic human, other or unspecified agent Giardiasis Gonorrhea Haemophilus influenzae, invasive disease Hansen disease (leprosy) Hantavirus pulmonary syndrome Hemolytic uremic syndrome, postdiarrheal Hepatitis A, acute Hepatitis B, acute Hepatitis B, chronic† Hepatitis B virus, perinatal infection Hepatitis C, acute† Hepatitis C virus infection (past or present) Human immunodeficiency virus (HIV) infection adult (age =13 yrs) pediatric (age <13 yrs) Influenza-associated pediatric mortality Legionellosis Listeriosis Lyme disease Malaria Measles§ Meningococcal disease Mumps Novel influenza A virus infections¶ Pertussis Plague Poliomyelitis, paralytic Poliovirus infection, nonparalytic¶ Psittacosis Q fever Rabies animal human Rocky Mountain spotted fever Rubella§ Rubella, congenital syndrome§ Salmonellosis Severe acute respiratory syndrome--associated coronavirus (SARS-CoV) disease Shiga toxin-producing Escherichia coli (STEC) Shigellosis Smallpox Streptococcal disease, invasive, group A Streptococcal toxic-shock syndrome Streptococcus pneumoniae, invasive disease** drug resistant, all ages age <5 years, nondrug resistant Syphilis Syphilis, congenital Tetanus Toxic-shock syndrome (other than streptococcal) Trichinellosis Tuberculosis Tularemia Typhoid fever Vancomycin-intermediate Staphylococcus aureus infection (VISA)† Vancomycin-resistant Staphylococcus aureus infection (VRSA)† Varicella infection (morbidity) Varicella (mortality) Vibriosis (non-cholera Vibrio infections)¶ Yellow fever Data SourcesProvisional data concerning the reported occurrence of nationally notifiable infectious diseases are published weekly in MMWR. After each reporting year, staff in state health departments finalize reports of cases for that year with local or county health departments and reconcile the data with reports previously sent to CDC throughout the year. These data are compiled in final form in the Summary. Notifiable disease reports are the authoritative and archival counts of cases. They are approved by the appropriate chief epidemiologist from each submitting state or territory before being published in the Summary. Data published in MMWR Surveillance Summaries or other surveillance reports produced by CDC programs might not agree exactly with data reported in the annual Summary because of differences in the timing of reports, the source of the data, or surveillance methodology. Data in the Summary were derived primarily from reports transmitted to CDC from health departments in the 50 states, five territories, New York City, and the District of Columbia. Data were reported for MMWR weeks 1--52, which correspond to the period for the week ending January 6, 2007, through the week ending December 29, 2007. More information regarding infectious notifiable diseases, including case definitions, is available at http://www.cdc.gov/ncphi/disss/nndss/nndsshis.htm. Policies for reporting notifiable disease cases can vary by disease or reporting jurisdiction. The case-status categories used to determine which cases reported to NNDSS are published, by disease or condition, and are listed in the print criteria column of the 2007 NNDSS event code list (available at http://www.cdc.gov/ncphi/disss/nndss/phs/files/NNDSS_event_code_list_January_2008.pdf). Final data for certain diseases are derived from the surveillance records of the CDC programs listed below. Requests for further information regarding these data should be directed to the appropriate program. Coordinating Center for Health Information and Service National Center for Health Statistics (NCHS) Office of Vital and Health Statistics Systems (deaths from selected notifiable diseases). Coordinating Center for Infectious Diseases National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention (NCHHSTP). Division of HIV/AIDS Prevention (AIDS and HIV infection) Division of STD Prevention (chancroid; Chlamydia trachomatis, genital infection; gonorrhea; and syphilis) Division of Tuberculosis Elimination (tuberculosis) National Center for Immunization and Respiratory Diseases Influenza Division (influenza-associated pediatric mortality). Division of Viral Diseases, (poliomyelitis, varicella [morbidity and deaths], and SARS-CoV). National Center for Zoonotic, Vector-Borne, and Enteric Diseases Division of Vector-Borne Infectious Diseases (arboviral diseases). Division of Viral and Rickettsial Diseases (animal rabies). Population estimates for the states are from the NCHS bridged-race estimates of the July 1, 2000--July 1, 2006 U.S. resident population from the vintage 2006 postcensal series by year, county, age, sex, race, and Hispanic origin, prepared under a collaborative arrangement with the U.S. Census Bureau. This data set was released on August 16, 2007, and is available at http://www.cdc.gov/nchs/about/major/dvs/popbridge/popbridge.htm. Populations for territories are 2006 estimates from the U.S. Census Bureau International Data Base, available at http://www.census.gov/ipc/www/idb/summaries.html. The choice of population denominators for incidence reported in the MMWR is based on 1) the availability of census population data at the time of preparation for publication and 2) the desire for consistent use of the same population data to compute incidence reported by different CDC programs. Incidence in the Summary is calculated as the number of reported cases for each disease or condition divided by either the U.S. resident population for the specified demographic population or the total U.S. residential population, multiplied by 100,000. When a nationally notifiable disease is associated with a specific age restriction, the same age restriction is applied to the population in the denominator of the incidence calculation. In addition, population data from states in which the disease or condition was not notifiable or was not available were excluded from incidence calculations. Unless otherwise stated, disease totals for the United States do not include data for American Samoa, Guam, Puerto Rico, the Commonwealth of the Northern Mariana Islands, or the U.S. Virgin Islands. Interpreting DataIncidence data in the Summary are presented by the date of report to CDC as determined by the MMWR week and year assigned by the state or territorial health department, except for the domestic arboviral diseases, which are presented by date of diagnosis. Data are reported by the state in which the patient resided at the time of diagnosis. For certain nationally notifiable infectious diseases, surveillance data are reported independently to different CDC programs. For this reason, surveillance data reported by other CDC programs might vary from data reported in the Summary because of differences in 1) the date used to aggregate data (e.g., date of report or date of disease occurrence), 2) the timing of reports, 3) the source of the data, 4) surveillance case definitions, and 5) policies regarding case jurisdiction (i.e., which state should report the case to CDC). The data reported in the Summary are useful for analyzing disease trends and determining relative disease burdens. However, reporting practices affect how these data should be interpreted. Disease reporting is likely incomplete, and completeness might vary depending on the disease and reporting state. The degree of completeness of data reporting might be influenced by the diagnostic facilities available; control measures in effect; public awareness of a specific disease; and the interests, resources, and priorities of state and local officials responsible for disease control and public health surveillance. Finally, factors such as changes in methods for public health surveillance, introduction of new diagnostic tests, or discovery of new disease entities can cause changes in disease reporting that are independent of the true incidence of disease. Public health surveillance data are published for selected racial/ethnic populations because these variables can be risk markers for certain notifiable diseases. Race and ethnicity data also can be used to highlight populations for focused prevention efforts. However, caution must be used when drawing conclusions from reported race and ethnicity data. Different racial/ethnic populations might have different patterns of access to health care, potentially resulting in data that are not representative of actual disease incidence among specific racial/ethnic populations. Surveillance data reported to NNDSS are in either individual case-specific form or summary form (i.e., aggregated data for a group of cases). Summary data often lack demographic information (e.g., race); therefore, the demographic-specific rates presented in the Summary might be underestimated. In addition, not all race and ethnicity data are collected uniformly for all diseases. For example, certain disease programs collect data on race and ethnicity using one or two variables, based on the 1977 standards for collecting such data issued by the Office of Management and the Budget (OMB). However, beginning in 2003, certain CDC programs, such as the tuberculosis program, implemented OMB's 1997 revised standards for collecting such data; these programs collect data on multiple races per person using multiple race variables. In addition, although the recommended standard for classifying a person's race or ethnicity is based on self-reporting, this procedure might not always be followed. Transition in NNDSS Data Collection and ReportingBefore 1990, data were reported to CDC as cumulative counts rather than individual case reports. In 1990, states began electronically capturing and reporting individual case reports (without personal identifiers) to CDC using the National Electronic Telecommunication System for Surveillance (NETSS). In 2001, CDC launched the National Electronic Disease Surveillance System (NEDSS), now a component of the Public Health Information Network, to promote the use of data and information system standards that advance the development of efficient, integrated, and interoperable surveillance information systems at the local, state, and federal levels. One of the objectives of NEDSS is to improve the accuracy, completeness, and timeliness of disease reporting at the local, state, and national level. CDC has developed the NEDSS Base System (NBS), a public health surveillance information system that can be used by states that do not have their own NEDSS-compatible based systems. A major feature of NBS is the ability to capture data already in electronic form (e.g., electronic laboratory results, which are needed for case confirmation) rather than enter these data manually as in NETSS. In 2007, 16 states used NBS to transmit nationally notifiable infectious diseases to CDC. Additional NBS information concerning NEDSS is available at http://www.cdc.gov/NEDSS. Change in Methodology for Identifying Which Nationally Notifiable Infectious Diseases Were Not Reportable in National Notifiable Diseases Surveillance System (NNDSS) Reporting Jurisdictions in 2007In 2007, the (NNDSS) program changed the methodology used to gather information regarding which nationally notifiable infectious diseases were reportable in U.S. states and territories. The NNDSS program provided technical assistance to the Council of State and Territorial Epidemiologists (CSTE) in implementing the CSTE State Reportable Conditions Assessment (SRCA). This assessment solicited information from each NNDSS reporting jurisdiction (all 50 U.S. states, the District of Columbia, New York City, and five U.S. territories) regarding which public health conditions were reportable by clinicians, laboratories, hospitals, or 'other" public health reporters, as mandated by law or regulation. A total of 255 conditions, including infectious conditions and noninfectious conditions (e.g., injuries, cancer, and work-related conditions) were included in the assessment. Information concerning all nationally notifiable diseases was also captured by the assessment. The 2007 assessment was the first collaborative project of such technical magnitude ever conducted by CSTE and CDC in which CDC and CSTE had previously gathered public health reporting requirements independently. The SRCA collected information regarding whether each reportable condition was explicitly reportable (i.e., listed as a specific disease or as a category of diseases on reportable disease lists) or whether it was implicitly reportable (i.e., included in a general category of the reportable disease list, such as "rare diseases of public health importance"), or not reportable. Only explicitly reportable conditions were considered reportable for the purpose of national public health surveillance, and thus NNDSS. Moreover, to determine whether a condition included in the SRCA was considered reportable across all public-health--reporter categories and for a specific NNID in a reporting jurisdiction, CDC developed and applied a condition algorithm and a results algorithm to run on the data collected in the 2007 SRCA. Analyzed results of the 2007 SRCA were used to determine whether a NNID was not reportable in a reporting jurisdiction in 2007 and thus noted with an "N" indicator (for "not notifiable") in the front tables of this report. Unanalyzed results from the 2007 SRCA (and a subsequent 2008 SRCA) are available, using CSTE's web-query tool, at http://www.cste.org/dnn/programsandacttivities/publichealthinformatics/statereportableconditionsqueryresults/tabid/261/default.aspx. Revised International Health RegulationsIn May 2005, the World Health Assembly adopted revised International Health regulations (IHR) (2) that went into effect in the United States on July 18, 2007. This international legal instrument governs the role of the World Health Organization (WHO) and its member countries, including the United States, in identifying, responding to and sharing information about Public Health Emergencies of International Concern (PHEIC). A PHEIC is an extraordinary event that 1) constitutes a public health risk to other countries through international spread of disease, and 2) potentially requires a coordinated international response. The IHR are designed to prevent and protect against the international spread of diseases while minimizing the effect on world travel and trade. Countries that have adopted these rules have a much broader responsibility to detect, respond to, and report public health emergencies that potentially require a coordinated international response in addition to taking preventive measures. The IHR will help countries work together to identify, respond to, and share information about PEHIC. The revised IHR represent a conceptual shift from a predefined disease list to a framework of reporting and responding to events on the basis of an assessment of public health criteria, including seriousness, unexpectedness, and international travel and trade implications. PHEIC are events that fall within those criteria (further defined in a decision algorithm in Annex 2 of the revised IHR). Four conditions always constitute a PHEIC and do not require the use of the IHR decision instrument in Annex 2: Severe Acute Respiratory Syndrome (SARS), smallpox, poliomyelitis caused by wild-type poliovirus, and human influenza caused by a new subtype. Any other event requires the use of the decision algorithm in Annex 2 of the IHR to determine if it is a potential PHEIC. Examples of events that require the use of the decision instrument include, but are not limited to, cholera, pneumonic plague, yellow fever, West Nile fever, viral hemorrhagic fevers, and meningococcal disease. Other biologic, chemical, or radiologic events might fit the decision algorithm and also must be reportable to WHO. All WHO member states are required to notify WHO of a potential PHEIC. WHO makes the final determination about the existence of a PHEIC. Health-care providers in the United States are required to report diseases, conditions, or outbreaks as determined by local, state, or territorial law and regulation, and as outlined in each state's list of reportable conditions. All health-care providers should work with their local, state, and territorial health agencies to identify and report events that might constitute a potential PHEIC occurring in their location. U.S. State and Territorial Departments of Health have agreed to report information about a potential PHEIC to the most relevant federal agency responsible for the event. In the case of human disease, the U.S. State or Territorial Departments of Health will notify CDC rapidly through existing formal and informal reporting mechanisms (3). CDC will further analyze the event based on the decision algorithm in Annex 2 of the IHR and notify the U.S. Department of Health and Human Services (DHHS) Secretary's Operations Center (SOC), as appropriate. DHHS has the lead role in carrying out the IHR, in cooperation with multiple federal departments and agencies. The HHS SOC is the central body for the United States responsible for reporting potential events to the WHO. The United States has 48 hours to assess the risk of the reported event. If authorities determine that a potential PHEIC exists, the WHO member country has 24 hours to report the event to the WHO. An IHR decision algorithm in Annex 2 has been developed to help countries determine whether an event should be reported. If any two of the following four questions can be answered in the affirmative, then a determination should be made that a potential PHEIC exists and WHO should be notified:
Additional information concerning IHR is available at http://www.who.int/csr/ihr/en, http://www.globalhealth.gov/ihr/index.html, http://www.cdc.gov/cogh/ihregulations.htm, and http://www.cste.org/PS/2007ps/2007psfinal/ID/07-ID-06.pdf. At its annual meeting in June 2007, the Council of State and Territorial Epidemiologists (CSTE) approved a position statement to support the implementation of the IHR in the United States (3). CSTE also approved a position statement in support of the 2005 IHR adding initial detections of novel influenza A virus infections to the list of nationally notifiable diseases reportable to NNDSS, beginning in January 2007 (4).
* No cases of diphtheria; neuroinvasive or nonneuroinvasive western equine encephalitis virus disease; poliomyelitis, paralytic; poliovirus infection, nonparalytic; rubella, congenital syndrome; severe acute respiratory syndrome--associated coronavirus syndrome, (SARS-CoV); smallpox; and yellow fever were reported in 2007; these conditions do not appear in the tables in Part 1. For certain other nationally notifiable diseases, incidence data were reported to CDC but are not included in the tables or graphs of this Summary. Data on chronic hepatitis B and hepatitis C virus infection (past or present) are undergoing quality review. Data on human immunodeficiency virus (HIV) infections are not included because HIV reporting has been implemented on different dates and using different methods than for AIDS case reporting. * Position Statements the Council of State and Territorial Epidemiologists (CSTE) approved in 2006 for national surveillance were implemented beginning in January 2007. † Revised national surveillance case definition. § Updated case classifications. ¶ Added to national notifiable disease list, 2007. ** New reporting guidelines. Highlights for 2007Below are summary highlights for certain national notifiable diseases. Highlights are intended to assist in the interpretation of major occurrences that affect disease incidence or surveillance trends (e.g., outbreaks, vaccine licensure, or policy changes). AIDSSince 1981, confidential name-based AIDS surveillance has been the cornerstone of national, state, and local efforts to monitor the scope and impact of the HIV epidemic. The data have multiple uses, including the development of policy to help prevent and control AIDS. However, because of the introduction of therapies that effectively slow the progression of human immunodeficiency virus (HIV) infection, AIDS data no longer adequately represent the populations affected by the epidemic. By helping public health practitioners understand the epidemic at an earlier stage, combined HIV and AIDS data better represent the overall impact of HIV. As of April 2008, all 50 states, the District of Columbia, and five U.S. territories had implemented confidential name-based HIV reporting. These areas have integrated name-based HIV surveillance into their AIDS surveillance systems; names or other personal indentifying information are not reported to CDC. At the end of 2007, an estimated 455,636 persons in the United States were living with AIDS (1). During 2003--2007, the estimated number of new AIDS cases decreased 7.5%, from 38,893 cases in 2003 to 35,962 in 2007. More than 1 million persons in the United States were estimated to have received an AIDS diagnosis from the beginning of the epidemic through the end of 2007.
AnthraxIn August 2007, two related cases of cutaneous anthrax, one confirmed and one probable, occurred in Connecticut. Exposure was determined to be the result of making traditional West African drums using untreated, Bacillus anthracis--contaminated goat hides that were imported from Guinea in the first case and cross-contamination of the residence by the drum maker in the second case. Both patients recovered with treatment (1). This event, and previous, unrelated cases of anthrax associated with contaminated animal skin drums (2,3), demonstrate the health risks that can be posed by drums made with hides imported from countries where anthrax is common. Persons who make, play or handle these drums, or who are exposed to environments cross-contaminated by the making of these drums, are potentially at risk. Naturally occurring anthrax epizootics continue to be reported annually among U.S. wildlife and livestock populations, with epizootics reported by six states in 2007, including California, North and South Dakota, Minnesota, Montana, and Texas.
Arboviral, Neuroninvasive and Nonneuroinvasive(West Nile virus)During 2007, West Nile virus (WNV) activity was detected in 47 states and expanded into 19 counties that had not reported WNV activity previously. Human WNV disease cases were reported in 44 states (1). Nationally, the reported incidence of West Nile neuroinvasive disease (WNND) was 0.4 cases per 100,000 population, which is similar to that reported in 2004 (0.4), 2005 (0.4), and 2006 (0.5). The relative stability in the number of reported WNND cases during the past 4 years likely represents endemic WNV transmission in the continental United States. The highest incidence of WNND continued to occur in western and central states. In 2007, human WNV infection was identified for the first time in Puerto Rico among three asymptomatic blood donors. BrucellosisSince 2003, incidence of brucellosis in the United States has increased yearly. During 2006--2007, the number of cases increased 12.1%. The overall characteristics of persons with brucellosis remain stable. For patients for whom ethnicity was identified, 63.5% were Hispanic. The majority of cases were reported in the Southwest. In the U.S. animal population, brucellosis eradication efforts continue. In 2007, the U.S. Department of Agriculture declared Idaho a brucellosis Class Free state. Texas remained a Class A state but reported no new infected cattle herds (1). In total, 49 states and three territories were classified as Brucellosis Class Free states at the end of 2007 (1). Risk factors associated with brucellosis include the consumption of unpasteurized milk or soft cheeses. The risk for brucellosis from dairy produced in the United States is low. Unpasteurized dairy from countries with endemic brucellosis remains a source of brucellosis for immigrants and travelers. Hunters are at an elevated risk, as Brucella abortus remains enzootic in elk and bison in the greater Yellowstone National Park area, and Brucella suis is enzootic in feral swine in the Southeast. In addition, exposure to Brucella spp. can occur accidentally in diagnostic and research laboratories because of a high potential for aerosol transmission (2). For the same reason, biosafety level 3 practices, containment, and equipment are recommended for laboratory manipulation of isolates (3). In the event of an exposure, postexposure prophylaxis can effectively prevent seroconversion and subsequent illness (4). CDC provides recommendations for laboratory exposures and can assist with the serological monitoring of exposed laboratory workers.
CholeraCases of cholera continue to be rare in the United States. The number of cases reported in 2007 was higher than the average number of cases per year reported during 2002--2006 (5.8) (1). Foreign travel continues to be the primary source of illness for cholera in the United States. Cholera remains a global threat to health, particularly in areas with poor access to improved water and sanitation, such as sub-Saharan Africa (2). All patients with domestic exposure in 2007 had consumed seafood. Other serogroups of cholera-toxin--producing Vibrio cholerae (e.g., O141 and O75) also have caused severe diarrhea in patients with a history of consumption of seafood from the Gulf Coast (3).
CoccidioidomycosisDuring 2007, the number of reported coccidioidomycosis cases in the United States decreased slightly, primarily because of a decrease in the number of reports received from the disease-endemic states of Arizona and, to a lesser extent, California. Coccidioidomycosis is a common cause of community-acquired pneumonia in disease-endemic areas, despite infrequent testing. In 2007, the Council of State and Territorial Epidemiologists adopted modifications to the previous surveillance case definitions (1). The revised case definition allows for a positive serologic test for IgG (any of several clinically accepted methods) without a confirmation of a rising IgG titer to be sufficient for case confirmation. As a result, case counts are expected to rise during 2008.
CryptosporidiosisIn 2007, the number of cryptosporidiosis cases reported increased dramatically. This follows an increase in the number of cases in 2005 and 2006. The reasons for this continued increase are unclear but might reflect changes in jurisdictional reporting patterns, increased testing for Cryptosporidium, or a real increase in infection and disease caused by Cryptosporidium. Cryptosporidiosis is widespread geographically in the United States (1), and during 2004--2007, incidence increased in every state. As in previous years, cryptosporidiosis case reports were influenced by outbreaks, particularly those associated with treated recreational water. Although cryptosporidiosis affects persons in all age groups, the number of reported cases was highest among children aged 1--9 years. A tenfold increase in transmission of cryptosporidiosis occurred during summer through early fall, coinciding with increased use of recreational water by younger children, which is a known risk factor for cryptosporidiosis. Cryptosporidium oocysts can be detected routinely in treated recreational water (2). Contamination of, and the subsequent transmission through, recreational water is facilitated by the substantial number of Cryptosporidium oocysts that can be shed by a single person; the extended periods of time that oocysts can be shed (3); the low infectious dose (4); the resistance of Cryptosporidium oocysts to chlorine (5); and the prevalence of improper pool maintenance (i.e., insufficient disinfection, filtration, and recirculation of water), particularly of children's wading pools (6).
Ehrlichiosis and AnaplasmosisHuman monocytic ehrlichiosis (also called HME or ehrlichiosis, caused by Ehrlichia chaffeensis) and human granulocytic ehrlichiosis (also called HGE or anaplasmosis, caused by Anaplasma phagocytophilum) have been reported since 1999. Cases reported for 2007 represent a 44% increase in HME and a 29% increase in HGE over those reported for 2006, and an overall 159% and 130% increase since 2003, respectively. Increases in the numbers of reported cases might be the result of several factors, including possible increases in vector tick populations, or increases in human/tick contact as a result of human encroachment into tick habitat through recreational activities and housing construction. In addition, artifactual increases in reported cases might relate to changes in surveillance techniques or perception/awareness of disease, as might occur through changes in case definitions (which occurred in 2000 and 2004), and the constant evolution of laboratory tests most commonly available for diagnosis. Expected geographic areas of occurrence for ehrlichiosis and anaplasmosis are based on known distributions of primary tick vectors responsible for infection. HME (ehrlichiosis) cases are expected to be reported primarily in the lower Midwest and the Southeast, reflecting the range of the primary tick vector species (Amblyomma americanum). HGE (anaplasmosis) cases are expected to be reported primarily from the upper Midwest and coastal New England, reflecting both the range of the primary tick vector species (Ixodes scapularis) and preferred animal hosts for tick feeding. However, human antibody responses to both of these infections often exhibit strong immunologic cross-reactivity using available serologic laboratory tests. In certain areas in which the range of expected tick vectors is known to overlap (e.g., in the central eastern United States) and for which one species is not differentiated by serologic testing, cases might be reported most appropriately as "Ehrlichiosis, unspecified." However, the large number of cases of "Human Monocytic Ehrlichiosis" and "Ehrlichiosis, unspecified" reported from the northeastern and upper midwestern United States more likely reflects situations in which physicians, confused as to causative agent, ordered single, incorrect tests resulting in incomplete diagnostic testing and interpretation (e.g., physicians ordering only HME tests in a region where HGE would be expected to predominate). Ehrlichia ewingii cases reflected in the "Ehrlichiosis, unspecified" category are impossible to ascertain, because only molecular diagnostic tests can be used to diagnose ehrlichiosis resulting from this infection; however, numbers are expected to be low. Since the 2007 reporting year, the case definitions for these diseases have been modified by a resolution adopted by the Council of State and Territorial Epidemiologists to include a separate designation for E. ewingii for better assessment and counting of cases; the new category names and the new case definitions became effective January 1, 2008 (1) and will be first reflected in the 2008 Notifiable Disease Summary epidemiology of gonorrhea.
Hansen Disease (Leprosy)The number of cases of Hansen disease (HD) reported in the United States peaked at 361 in 1985 and decreased from 1988 until 2006. In 2007, the number of cases reported to CDC increased by 49% from the number reported in 2006; this increase might be attributable to CDC attempts made to improve reporting. Cases were reported from 25 states and two U.S. territories; 69% of cases were reported from Texas, Hawaii, Florida, California, New York City, and Guam. HD is not highly transmissible; cases appear to be related predominantly to immigration from areas in which the disease is endemic. More information on access to clinical care is available at http://www.hrsa.gov/hansens. Hemolytic Uremic Syndrome, PostdiarrhealHemolytic uremic syndrome (HUS) is characterized by the triad of hemolytic anemia, thrombocytopenia, and renal insufficiency. The most common etiology of HUS in the United States is infection with Shiga toxin--producing Escherichia coli, principally E. coli O157:H7 (1). Approximately 8% of persons infected with E. coli O157:H7 progress to HUS (2). During 2007, as usual, the majority of reported cases occurred among children aged 1--4 years.
Viral Hepatitis, AcuteImplementation of the 1999 recommendations for routine childhood hepatitis A vaccination in the United States has reduced rates of infection; universal vaccination of children against hepatitis B also has reduced disease incidence substantially among younger age groups (1). Higher rates of hepatitis B virus (HBV) infections continue among adults, particularly men aged 25 -- 44 years, reflecting the need to vaccinate adults at risk for HBV infection (e.g., injection-drug users [IDUs] and men who have sex with men). Although screening of blood after 1992 for hepatitis C antibodies has been important, the decline in hepatitis C incidence that has occurred in the past decade is attributable primarily to a decrease in incidence among IDUs (2). The reasons for this decrease are unknown but likely reflect changes in behavior and practices among this population. In 2006, the expansion of recommendations for routine hepatitis A vaccination to include all children in the United States aged 12-- 23 months is expected to reduce hepatitis A rates further. Ongoing hepatitis B vaccination programs ultimately will eliminate domestic HBV transmission, and increased vaccination of adults with risk factors will accelerate progress toward elimination. Prevention of hepatitis C relies on identifying and counseling uninfected persons at risk for hepatitis C (e.g., IDUs) regarding ways to protect themselves from infection and on identifying and preventing transmission of HCV in health-care settings (3).
HIV Infection, AdultSince 2004, a total of 39 areas (34 states and five dependent areas) have had laws or regulations requiring confidential name-based reporting for human immunodeficiency virus (HIV) infection, in addition to reporting of persons with AIDS. In 2002, CDC initiated a system to monitor HIV incidence; in 2003 this system was expanded. On the basis of extrapolations for the 22 states with HIV incidence surveillance, the estimated number of new HIV infections for the United States in 2006 was 56,3000 (1). At the end of 2007, an estimated total of 261,741 adults and adolescents in the 39 areas were living with HIV infection (not AIDS) (2). The estimated prevalence rate of HIV infection (not AIDS) in this group was 154.2 per 100,000 population. In these areas, 2007 was the first year in which mature HIV surveillance data (i.e., data available since at least 2003) could be used to allow for stabilization of data collection and for adjustment of the data for reporting delays. Data from additional areas will be included in analyses when =4 years of HIV case reports have accrued.
HIV Infection, PediatricAt the end of 2007, in the 39 areas (34 states and five dependent areas) that have had confidential name-based human immunodeficiency virus (HIV) surveillance since at least 2003 for children aged <13 years with confirmed HIV infection, an estimated 2,195 children were living with HIV infection (not AIDS) (1). Estimated prevalence of HIV infection (not AIDS) in this group was 6.0 per 100,000 population.
Influenza-Associated Pediatric MortalityAn early and severe influenza season during 2003--2004 was associated with deaths in children in several states, prompting CDC to request that all state, territorial, and local health departments report laboratory-confirmed influenza-associated pediatric deaths in children aged <18 years (1,2). During the 2003--04 influenza season, 153 pediatric influenza-associated deaths were reported to CDC by 40 state health departments (3). In June 2004, the Council of State and Territorial Epidemiologists added influenza-associated pediatric mortality to the list of conditions reportable to the National Notifiable Diseases Surveillance System (4). Cumulative year-to-date incidence is published each week in MMWR Table I for low-incidence nationally notifiable diseases. The number of influenza associated pediatric deaths reported to CDC increased during 2007 compared with 2006 (77 and 43, respectively). The median age at death was 7.4 years (range: 9 days---17.8 years) and was higher than in 2006 (median age: 4 years). In 2007, a total of 13 children (17%) were aged <6 months; 10 (13%) were aged 6--23 months; 10 (13%) were aged 24--59 months; and 44 (57%) were aged >5 years. In 2007, of the 76 children for whom location at the time of death was reported, 42 (55%) died after being admitted to the hospital, whereas 34 (45%) died in the emergency room or outside the hospital. The percentage of children who died in the emergency room or outside the hospital was similar to that reported during 2006 (47%). Information on underlying or chronic medical conditions was reported for 72 children: 31 (43%) children had one or more underlying or chronic conditions, and 41 (57%) were previously healthy. The more common chronic conditions reported included moderate to severe developmental delay (n = 11), seizure disorder (n = nine), asthma (n = seven), cardiac disease (n = six), and a history of febrile seizures (n = four). Invasive bacterial coinfection was reported in 28 (36%) children. This represents an increase compared with 2006, when seven (16%) of 43 children had bacterial coinfections. Staphylococcus aureus was the most frequently reported bacterial pathogen in 2007 and was found in 19 (68%) of the 28 children with coinfections. Of the 19 Staphylococcus isolates, 13 were methicillin-resistant, four were methicillin-sensitive, and for two, sensitivity testing was not performed. Other bacterial pathogens identified included Group A Streptococcus (n = four), Group B Streptococcus (n = one), Klebseilla pneumoniae (n = one), Haemophilus influenzae (n = one), Acinebactor (n = one), Enterobacter cloacae (n = one), Haemophilus influenzae (n = one), and Nocardia (n = one). Two children had two bacterial pathogens identified. Data on influenza vaccination status were reported for 71 children: six children (8%) received at least 1 dose of influenza vaccine before the onset of illness during 2007, and only four (6%) were fully vaccinated. The current recommendations of CDC's Advisory Committee on Immunization Practices (ACIP) (5) include annual vaccination of all children aged 6 months--18 years. Annual vaccination of all children aged 5--18 years should begin in September or as soon as vaccine is available for the 2008--09 influenza season, if feasible, but annual vaccination of all children aged 5--18 years should begin no later than during the 2009--10 influenza season. Administration of 2 doses of influenza vaccine is necessary for previously unvaccinated children aged 6 months--<9 years. Continued surveillance of severe influenza-related mortality is important to monitor the effect of influenza and the possible effects of interventions in children.
LegionellosisTotal legionellosis case reports and incidence for the past 5 years have been consistently higher than earlier years. During 2007, a slight decline occurred in the number of reported legionellosis cases compared with 2006. Compared with 2006, the Mid-Atlantic region had the greatest decline in reported cases in 2007 (14%), whereas the West South Central region, specifically Texas, had the greatest increase in reported cases (63%). Though approximately 40% of reported cases occur in persons aged = 65 years, pediatric cases (aged = 18 years) were reported and accounted for approximately 1% of cases. Although legionellosis is less common in the pediatric population than among adults, many cases are likely undiagnosed. As in previous years, the majority of reported cases were sporadic; of those with known outbreak status, less than 1% were reported to be associated with an outbreak. ListeriosisListeriosis is a rare but severe infection caused by Listeria monocytogenes; it has been a nationally notifiable disease since 2000. Listeriosis is primarily foodborne and occurs most frequently among persons who are older, pregnant, or immunocompromised. During 2007, the majority of cases occurred among persons aged =65 years. Molecular subtyping of L. monocytogenes isolates and sharing of that information through PulseNet has enhanced the ability of public health officials to detect and investigate outbreaks. Recent outbreaks have been linked to ready-to-eat deli meat (1) and unpasteurized cheese (2). During 2007, the incidence of listeriosis in FoodNet active surveillance sites was 0.27 cases per 100,000 population, representing a decrease of 42% compared with 1996--1998; however, the incidence remained higher than at its lowest point in 2002 (3). All clinical isolates should be submitted to state public health laboratories for pulsed-field gel electrophoresis (PFGE) pattern determination, and all persons with listeriosis should be interviewed by a public health official or health-care provider using a standard Listeria case form, available at http://www.cdc.gov/nationalsurveillance/PDFs/ListeriaCaseReportFormOMB0920-0004.pdf. Rapid analysis of surveillance data will allow identification of possible food sources of outbreaks.
Lyme DiseaseLyme disease is caused in North America by Borrelia burgdorferi sensu stricto, a spirochete transmitted by infected Ixodes ticks. Manifestations of infection include erythema migrans, arthritis, carditis, and neurologic deficits. During 2006--2007, the number of reported cases increased 38%, reaching 15% higher than the previous maximum reported in 2002. Much of this increase can be attributed to enhanced surveillance although evidence of true emergence exists in certain areas. The risk for Lyme disease can be reduced by avoiding tick-infested areas, using insect repellent containing N, N-diethyl-m-toluamide (DEET), and checking daily for attached ticks. The abundance of ticks around the home can be reduced through landscape modification and the use of area-wide acaricides (1,2).
MeaslesAs in recent years, 95% of confirmed measles cases in 2007 were import-associated. Of these, 29 cases were internationally imported and 12 resulted from exposure to persons with imported infections (1). The sources for the remaining two cases were classified as unknown because no link to importation was detected. More than half of all cases for 2007 occurred among adults aged >20 years; four persons were aged >50 years. Thirty-five percent of all cases occurred in U.S. residents who acquired infection while outside the United States, 80% of whom had either never been vaccinated with measles-containing vaccine or had unknown vaccination histories. Four outbreaks occurred during 2007 (range: 3--7 cases), all from imported sources. One outbreak associated with an international sporting event resulted in transmission to seven patients in three states, including one airline passenger and an airport worker (2). Although the elimination of endemic measles in the United States has been achieved, and population immunity remains high (3), outbreaks can occur when measles is introduced into susceptible groups, often at substantial cost to control (4). Measles can be prevented by adhering to recommendations for vaccination, including guidelines for travelers (5,6).
Meningococcal Disease, InvasiveNeisseria meningitidis is a leading cause of bacterial meningitis and sepsis in the United States. Rates of meningococcal disease are highest among infants, with a second peak at age 18 years (1). Rates of meningococcal disease are the lowest they have been in the United States, but meningococcal disease continues to cause substantial morbidity and mortality in all ages. A tetravalent (A,C,Y,W-135) meningococcal conjugate vaccine ([MCV4] Menactra(r) (Sanofi Pasteur, Swiftwater, Pennsylvania) is licensed for persons aged 2--55 years. In 2007, CDC's Advisory Committee on Immunization Practices revised recommendations for routine use of MCV4 to include children aged 11--12 years at the preadolescent vaccination visit and adolescents aged 13--18 years at the earliest opportunity (2). MCV4 also is recommended for college freshmen living in dormitories and other populations aged 2--55 years at increased risk for meningococcal disease (1). Further reductions in meningococcal disease could be achieved with the development of an effective serogroup B vaccine.
MumpsSince mumps vaccine licensure in 1967, the number of cases of mumps in the United States declined steadily until 2006, when the largest mumps outbreak in >20 years occurred, with >6,000 cases reported (1--4). In response to the 2006 outbreak, CDC's Advisory Committee on Immunization Practices (ACIP) updated criteria for mumps immunity and mumps vaccination recommendations (5). Acceptable presumptive evidence of immunity to mumps includes one of the following: 1) documentation of adequate vaccination, 2) laboratory evidence of immunity, 3) birth before 1957, or 4) documentation of physician-diagnosed mumps. Documentation of adequate vaccination now requires 2 doses of a live mumps virus vaccine for school-aged children (grades K--12) and adults at high risk (i.e., persons who work in health-care facilities, international travelers, and students at post--high school educational institutions). Health-care workers born before 1957 without other evidence of immunity should now consider 1 dose of live mumps vaccine. During an outbreak, a second dose of live mumps vaccine should be considered for children aged 1--4 years and adults at low risk if affected by the outbreak; health-care workers born before 1957 without other evidence of immunity should strongly consider 2 doses of live mumps vaccine. In 2007, the Council of State and Territorial Epidemiologists revised the mumps case definition for surveillance to be implemented January 1, 2008 (6).
Novel Influenza A VirusIn 2007, the Council of State and Territorial Epidemiologists adopted a position statement to include human infection with influenza A viruses that are different from currently circulating human influenza A(H1) and A(H3) as a reportable condition (1). Novel subtypes include, but are not limited to, completely different hemagglutinin glycoproteins such as H2, H5, H7, and H9, or influenza H1 and H3 subtypes originating from a nonhuman species or from genetic reassortment between animal and human viruses (2). In 2007, human cases of novel influenza A infection were reported from three states (Ohio, Illinois, and Michigan). Ill patients were infected with swine influenza A viruses: (swine H1N1 and H1N2). Both the swine H1N1 and H1N2 viruses were triple reassortants containing genes from swine, avian, and human viruses; both have been detected in recent years during outbreaks of respiratory illness in swine herds. Transmission of swine influenza viruses to humans usually occurs among persons in direct contact with ill pigs or persons who have been in places where pigs might have been present (e.g. agricultural fairs, farms, or petting zoos). Infected persons had either direct exposure to sick pigs or indirect exposure to a setting in which sick pigs were present. These cases, as with the other sporadic cases identified in recent years, have not resulted in sustained human-to-human transmission or community outbreaks.
PertussisIn 2007, the incidence of reported cases of pertussis (3.62 cases per 100,000 population) continued to decline after peaking during 2004--2005 at 8.9 cases per 100,000 population. Infants aged <6 months, who are too young to be fully vaccinated, had the highest reported rate of pertussis (69.9 cases per 100,000 population), but adolescents aged 10--19 years and adults aged >20 years contributed the greatest number of reported cases. Adolescents and adults might be a source of transmission of pertussis to young infants, who are at higher risk for severe disease and death and are recommended to be vaccinated with tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis vaccine (Tdap) (1,2). Although coverage with Tdap in adolescents aged 13--17 years increased during 2006--2007, from 10.8% to 30.4%, whether increased Tdap coverage has had an effect on disease rates is unknown (3).
Poliomyelitis, Paralytic and Poliovirus Infections, NonparalyticSince January 2007, in addition to paralytic poliomyelitis, nonparalytic poliovirus infections have been included in the list of nationally notifiable diseases reported through the National Notifiable Diseases Surveillance System (1,2). This addition resulted from the identification in 2005 of a type 1 vaccine-derived poliovirus (VDPV) infection among unvaccinated Minnesota children from a closed religious community who were not paralyzed (3). VDPV and wild polioviruses are still circulating worldwide and can be imported into the United States via U.S. or foreign-national travelers to these areas (4). Oral polio virus vaccine remains in widespread use globally and can be excreted in healthy and immunocompromised vaccinated persons. In 2005, a case of contact-acquired vaccine-associated paralytic poliomyelitis (VAPP) acquired overseas, occurred in an unvaccinated U.S. young adult (5). Public health officials should remain alert that paralytic poliomyelitis or poliovirus infections might occur in high-risk (i.e., unvaccinated or undervaccinated) populations and should report any detected poliovirus infections attributed to either wild or vaccine-derived polioviruses and any paralytic poliomyelitis cases.
PsittacosisHuman infection with Chlamydophila psittaci (psittacosis) is a potentially severe respiratory illness that occurs through exposure to infected birds. In 2007, the incidence of reported psittacosis cases continued to be low, decreasing slightly compared with 2006. The majority of cases occurred among women aged 25--49 years. Additional information regarding psittacosis, including case reporting tools, is available at http://www.nasphv.org/documentsCompendiaPsittacosis.html. Q FeverQ fever (caused by Coxiella burnetii) has been reported since 1999. Cases of Q fever reported for 2007 were similar to those reported for 2006, with a decrease of 9%. Despite this year's slight decrease, the overall number of cases reported in 2007 has increased 115% percent since 2003. In 2007, cases were distributed across the United States, in keeping with the consideration that Q fever is considered enzootic in ruminants (sheep, goats, and cattle) throughout the country. Although relatively few human cases are reported annually, the disease is believed to be substantially underreported because of its nonspecific presentation and the subsequent failure to suspect infection and request appropriate diagnostic tests. The 2007 reporting strategy does not differentiate acute Q fever infection from the more serious, life-threatening chronic form of infection, typically associated with endocarditis. The case definition for Q fever has been modified by a resolution adopted by the Council of State and Territorial Epidemiologists to include separate designations for acute and chronic forms of infection; the new category names and new case definitions became effective January 1, 2008 (1,2) and will be reflected in the 2008 Notifiable Disease Summary.
RabiesOne case of human rabies was reported during 2007 in a male aged 46 years from Minnesota. Epidemiologic investigation of this case implicated exposure to a bat as the most likely source of infection. As in previous years, the majority of animal rabies cases were reported in wild animal species. In the United States, five animal species are recognized as reservoir species for various rabies virus variants over defined geographic regions: raccoons (eastern United States.), bats (various species, all U. S. states except Hawaii), skunks (north central United States, south central United States, and California), foxes (Alaska, Arizona, and Texas), and mongoose (Puerto Rico) (1). Reported cases of rabies in domestic animals remain low, in part as a result of high vaccination rates and the elimination of dog-to-dog rabies transmission, which has not been reported in 3 years. One case of canine rabies imported in a dog from India was reported during 2007 (2). This case represents the continued challenge to the United States to remain canine-rabies--free. As in the past decade, cats were the most commonly reported domestic animal with rabies during 2007. Vaccination programs to control rabies in wild carnivores are ongoing through the distribution of baits containing an oral rabies vaccine in the eastern United States and Texas. Oral rabies vaccination programs in Texas are being maintained as a barrier to prevent the reintroduction of canine rabies from Mexico and to eliminate rabies in gray foxes in west Texas. Oral rabies vaccination programs also are being conducted in the eastern United States in an attempt to stop the westward spread of the raccoon rabies virus variant. In addition to oral rabies vaccination programs and routine public health activities (e.g., companion animal vaccination), public health education should target health-care providers and the public regarding rabies exposure prevention and postexposure prophylaxis recommendations.
Rocky Mountain Spotted FeverAfter several years in which case numbers were observed to have risen more than twofold, the 2007 reporting year for Rocky Mountain spotted fever (RMSF) caused by Rickettsia rickettsii appeared similar to 2006, decreasing only 3%. Cases in 2007 were distributed across the United States, reflecting the endemic status of RMSF and the widespread ranges of the primary tick vectors responsible for transmission (primarily Dermacentor variabilis and Dermacentor andersonii). RMSF cases associated with transmission by Rhipicephalus sanguineus, first reported in 2004, continued to be associated with cases reported from Arizona during 2007. (1). The overall 103% increase in the number of reported RMSF cases in the United States during the past 5 years might result from several factors, including possible increases in vector tick populations or increases in human/tick contact as a result of human encroachment into tick habitat through recreational activities and housing construction. In addition, artifactual increases in reported cases might reflect changes in surveillance techniques or perception/awareness of disease, as might occur through changes in case definitions (which occurred in 2004), increased recognition and reporting of illness that might be associated with genetically distinct rickettsial species incorrectly diagnosed as RMSF, and the constant evolution of laboratory tests most commonly available for diagnosis. The case definition for RMSF has been modified by a resolution adopted by the Council of State and Territorial Epidemiologists to include more specific information on interpretation of diagnostic laboratory criteria; the new case definitions became effective January 1, 2008 (2) and will be first reflected in the 2008 Summary of Notifiable Diseases.
SalmonellosisDuring 2007, as in previous years, proportionately the majority of cases of salmonellosis occurred among persons aged <5 years. Since 1993, the most frequently reported isolates have been Salmonella enterica serotype Typhimurium and S. enterica serotype Enteritidis (1). The epidemiology of Salmonella has been changing during the past decade. Salmonella serotype Typhimurium has decreased in incidence, whereas the incidence of serotypes Newport, Mississippi, and Javiana have increased. Specific control programs might have led to the reduction of serotype Enteritidis infections, which have been associated with the consumption of internally contaminated eggs. Rates of antibiotic resistance among several serotypes have been increasing; a substantial proportion of serotypes Typhimurium and Newport isolates are resistant to multiple drugs (2). The epidemiology of Salmonella infections is based on serotype characterization, and in 2005, the Council of State and Territorial Epidemiologists adopted a position statement for serotype-specific reporting of laboratory-confirmed salmonellosis cases (3).
Shiga Toxin-Producing Escherichia coli (STEC)Escherichia coli O157:H7 has been nationally notifiable since 1994 (1). National surveillance for all Shiga toxin-producing E. coli (STEC), under the name enterohemorrhagic E. coli (EHEC), began in 2001. In 2006, the nationally notifiable diseases case definition designation was changed from EHEC to STEC, and serotype-specific reporting was implemented (2). Diagnosis solely on the basis of detection of Shiga toxin does not protect public health sufficiently; characterizing STEC isolates by serotype and pulsed-field gel electrophoresis (PFGE) patterns is important to detect, investigate, and control outbreaks. Screening of stool specimens by clinical diagnostic laboratories for Shiga toxin by enzyme immunoassay with simultaneous bacterial culture using sorbitol-MacConkey agar (SMAC) is important for public health surveillance for STEC infections (3). All STEC isolates and enrichment broths from Shiga toxin-positive specimens that do not yield STEC O157 should be forwarded to state or local public health laboratories for further testing. Healthy cattle, which harbor the organism as part of the bowel flora, are the main animal reservoir of STEC. The majority of reported outbreaks are caused by contaminated food or water. The substantial decline in STEC cases reported during 2002--2003 coincided with industry and regulatory control activities and with a decrease in the contamination of ground beef (4). However, during 2005--2007, incidence of human STEC infections increased. Reasons for the increases are not known.
ShigellosisDuring 1978--2003, the number of shigellosis cases reported to CDC consistently exceeded 17,000. The approximately 14,000 cases of shigellosis reported to CDC in 2004 represented an all-time low. This number increased to approximately 16,000 in 2005, decreased slightly in 2006, and increased to almost 20,000 in 2007. Shigella sonnei infections continue to account for >75% of shigellosis in the United States (1). The majority of cases occur among young children, and large daycare-associated outbreaks are common and difficult to control (2). Certain cases are acquired during international travel (3,4). In addition to spread from one person to another, Shigellae can be transmitted through contaminated foods, sexual contact, and water used for drinking or recreational purposes (1). Resistance to ampicillin and trimethoprim-sulfamethoxazole among S. sonnei strains in the United States remains common (5).
Streptococcus pneumoniaeCases of invasive pneumococcal disease among children aged <5 years (IPD <5) and invasive drug-resistant Streptococcus pneumoniae (DRSP) among all ages are reportable to the National Notifiable Diseases Surveillance System (NNDSS) (1). The Council of State and Territorial Epidemiologists recommends reporting to track the impact of pneumococcal vaccination and other prevention programs. In 2007, several reporting areas did not require reporting of IPD <5 and/or DRSP. Among the 52 reporting areas, two states did not require reporting of IPD <5, three areas did not require reporting of DRSP, and seven areas did not require reporting of either type of case. Among those with a reporting requirement, two (5%) of 43 areas reported no cases of IPD <5, and 11 (26%) of 42 areas reported no cases of DRSP. Currently, data reported to the national level do not enable accurate assessment of pneumococcal disease burden or evaluation of immunization program impact. A new, 13-valent pneumococcal conjugate vaccine is expected to be licensed in 2009, and tracking cases of invasive pneumococcal disease will be important to monitor the effects of the vaccine on target populations (2). State-based surveillance systems and reporting should be enhanced now to provide baseline data and enable assessment of the impact of the new pneumococcal vaccine. A method for PCR-based serotyping is now available for use by state public health laboratories (3). Adopting this method provides an opportunity for state health departments to specifically track changes in serotypes targeted by the conjugate vaccines and would enhance their surveillance systems for vaccine-preventable IPD.
Syphilis, Primary and SecondaryIn 2007, primary and secondary (P&S) syphilis cases reported to CDC increased for the seventh consecutive year (1). During 2006--2007, the number of P&S syphilis cases reported to CDC increased 17.5%. P&S syphilis increased in all race/ethnicities except Asian/Pacific Islanders, but particularly among non-Hispanic blacks and Hispanics. Overall, increases in rates during 2001--2007 were observed primarily among men. In 2007, nearly two thirds (65%) of all P&S syphilis cases in 44 states and Washington D. C. (for which demographic information was available) occurred among men who have sex with men (MSM); 41% of MSM with P&S syphilis were white non-Hispanic, 33% were black non-Hispanic, and 19% were Hispanic. After decreasing during 1990--2004, the rate of P&S syphilis in women increased from 0.8 cases per 100,000 population in 2004 to 1.1 cases per 100,000 population in 2007. These increases occurred primarily in the South.
Syphilis, CongenitalDuring 2006--2007, after 14 years of decline, the rate of congenital syphilis increased 15.4% (1). This increase might relate to the increase in P&S syphilis among women that has occurred in recent years. In 2007, a total of 29 states and the District of Columbia had rates of congenital syphilis that exceeded the HP 2010 target of one case per 100,000 live births. The South accounted for 52% of these cases. The rate of congenital syphilis among non-Hispanic backs was 14 times higher than the rate among non-Hispanic whites, and the rate of congenital syphilis among Hispanics was almost seven times higher than the rate among non-Hispanic whites.
Typhoid FeverDespite recommendations that travelers to countries in which typhoid fever is endemic should be vaccinated with either of two effective vaccines available in the United States, approximately three fourths of all cases occur among persons who reported international travel during the preceding month and had not been vaccinated. Persons visiting friends and relatives in South Asia appear to be at particular risk, even during short visits (1). Recent illnesses have been attributed to ciprofloxacin-resistant isolates (CDC, unpublished data, 2007). Salmonella Typhi strains with decreased susceptibility to ciprofloxacin are increasingly frequent in that region and might require treatment with alternative antimicrobial agents (2,3). Although the number of S. Typhi infections is decreasing, the number of illnesses attributed to Salmonella Paratyphi A, which causes an illness indistinguishable from that caused by S. Typhi, is increasing. In a cross-sectional laboratory-based surveillance study conducted by CDC, 80% of patients with paratyphoid fever acquired their infections in South Asia, and 75% were infected with nalidixic acid-resistant strains, indicating decreased susceptibility to ciprofloxacin. A vaccine for paratyphoid fever is urgently needed (4).
Varicella (Chickenpox) DeathsThe Council of State and Territorial Epidemiologists recommended that deaths related to varicella be reportable to CDC starting in 1999 (1) as a first step towards national varicella surveillance. Although the number of varicella-related deaths has declined substantially since vaccine licensure from an annual average of 100--150 deaths (2), varicella-related deaths still occur. Of the six varicella-related deaths that were reported in 2007, five occurred in adults aged 23--78 years. More deaths were reported in 2007 than in 2006 (no deaths) or 2005 (three deaths) (3). However, because of incompleteness in reporting, the annual number of deaths is likely to be underestimated. Of the six deaths in 2007, one occurred in the context of an outbreak in a group mental health home. A male aged 35 years with unknown varicella vaccination and unknown disease history had severe sepsis after varicella infection and died 1 week after rash onset. Closed settings such as group homes are at risk for outbreaks of varicella-zoster virus (VZV) because such settings facilitate VZV transmission. This death highlights the importance of ensuring that both residents and employees of residential homes have evidence of immunity to VZV (4). One death occurred in a previously healthy girl aged 13 months who, although eligible for varicella vaccination, had not yet received the vaccine. After contracting varicella from her father, she had severe VZV infection with septic shock and died from multiple organ failure. The remaining four deaths occurred in adults with underlying medical conditions. One of these deaths occurred in an unvaccinated adult with no prior disease history. The varicella vaccination and disease history was unknown for the other three deaths.
VibriosisCholera, which is caused by infection with toxigenic Vibrio cholerae O1 and O139, has been nationally notifiable for many years. Infections attributable to other Vibrio species (vibriosis), especially V. parahaemolyticus and V. vulnificus, are a substantial public health burden. Infections are either foodborne or associated with wounds exposed to waters containing Vibrio species. In January 2007, vibriosis became a nationally notifiable disease reportable to the National Notifiable Diseases Surveillance System (NNDSS) (1). In addition to reporting through NNDSS, CDC requests that states collect information on the standard surveillance form for cholera and other Vibrio illness surveillance (available at http://www.cdc.gov/nationalsurveillance/cholera_vibrio_surveillance.html).
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. PART 1Summaries of Notifiable Diseases in the United States, 2007Abbreviations and Symbols Used in TablesU Data not available. N Not notifiable (i.e., report of disease is not required in that jurisdiction). --- No reported cases. Notes: Rates <0.01 after rounding are listed as 0. Data in the MMWR Summary of Notifiable Diseases --- United States, 2007 might not match data in other CDC surveillance reports because of differences in the timing of reports, the source of the data, and the use of different case definitions.
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. PART 2Graphs and Maps for Selected Notifiable Diseases in the United States, 2007Abbreviations and Symbols Used in Graphs and MapsU Data not available. N Not notifiable (i.e., report of disease not required in that jurisdiction). AS American Samoa CNMI Commonwealth of Northern Mariana Islands GU Guam PR Puerto Rico VI U.S. Virgin Islands AQUIRED IMMUNODEFICIENCY SYNDROME (AIDS). Percentage of reported cases, by race/ethnicity* --- United States, 2007 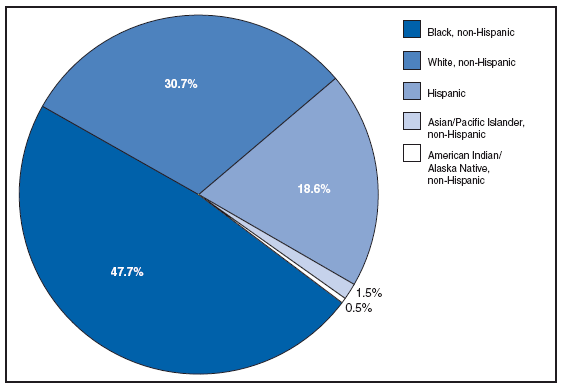
* For 1.0% of respondents, race was unknown. Of persons reported with AIDS during 2007, the greatest percentage were non-Hispanic blacks, followed by non-Hispanic whites, Hispanics, Asians/Pacific Islanders, and American Indians/Alaska Natives. AQUIRED IMMUNODEFICIENCY SYNDROME (AIDS). Reported AIDS rates* --- United States† and U.S. territories, 2007 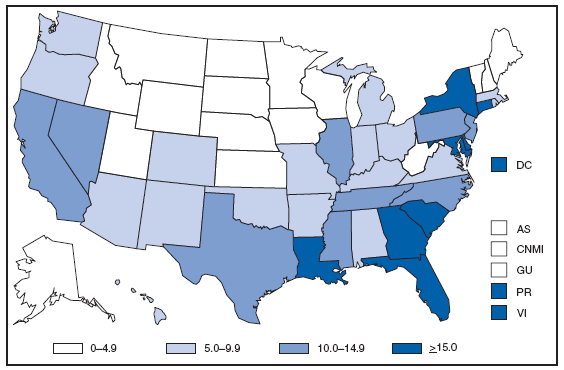
* Per 100,000 population. † Includes 222 persons with unknown state of residence. The highest AIDS rates were observed in the northeastern part of the country. High rates (i.e., =15 cases per 100,000 population) also were reported in several southeastern states, the U.S. Virgin Islands, and Puerto Rico. AQUIRED IMMUNODEFICIENCY SYNDROME (AIDS). Number of reported pediatric cases* -- United States† and U.S. Territories, 2007 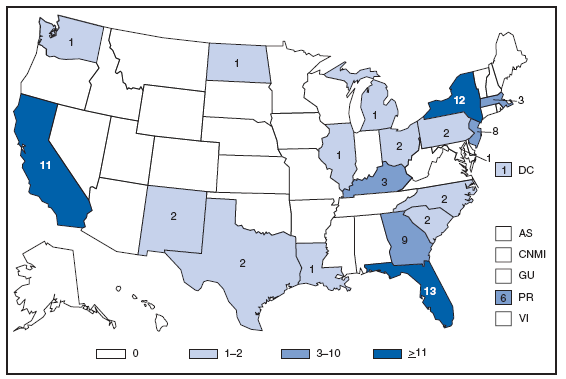
*Children and adolescents aged < 13 years. † Includes three persons with unknown state of residence. During 2007, a total of 87 new pediatric AIDS cases were reported in the United States and U.S. territories. Anthrax. Number of reported cases, by year -- United States, 1952--2007 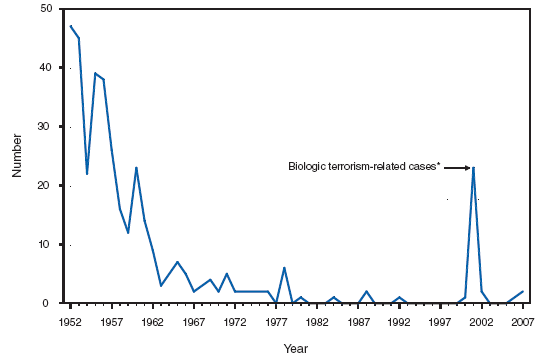
*One epizootic-associated cutaneous case was reported in 2001 from Texas. The two cases of cutaneous anthrax, one confirmed and one probable, that were reported in the United States during 2007 reflect the potential risk for anthrax among persons who make drums using untreated hides from countries where anthrax is common in animals and among persons who are exposed to environments that are cross-contaminated by these activities as a result of using untreated hides. This previously unrecognized source of risk for anthrax was first reported in 2006 as a result of two unrelated anthrax cases in the United States and United Kingdom, one of which was fatal, that occurred in persons exposed to making and playing animal-skin drums contaminated with Bacillus anthracis. Botulism, foodborne. Number of reported cases, by year --- United States, 1987--2007 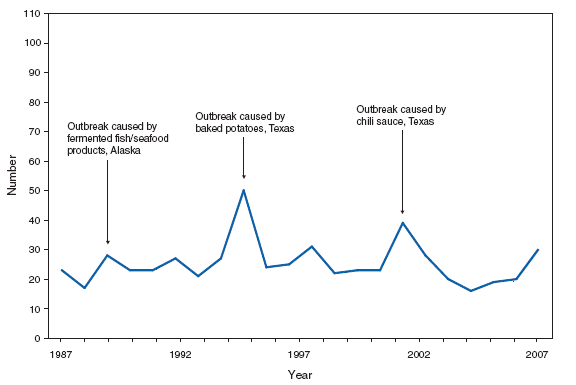
Home-canned foods as well as fermented foods commonly eaten by Alaska Native populations remain the principal sources of foodborne botulism in the United States. In 2007, a multistate outbreak of foodborne botulism was linked to commercially canned hotdog chili sauce. Botulism, infant. Number of reported cases, by year --- United States, 1987--2007 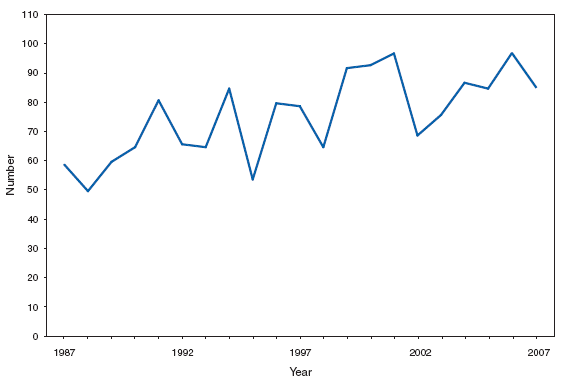
Infant botulism is the most common type of botulism in the United States. Cases are sporadic. Honey is the only known risk factor to date, though many cases have no known honey exposure. Botulism, other (includes wound and unspecified). Number of reported cases, by year --- United States, 1997--2007 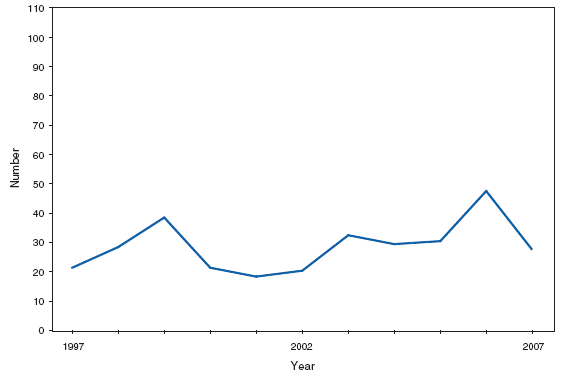
Wound botulism cases occur almost exclusively among injection-drug users in the western United States and are associated with a particular type of heroin known as black-tar heroin. The number of cases attributed to wound botulism was highest in 2006. Brucellosis. Number of reported cases, by year --- United States, 1977--2007 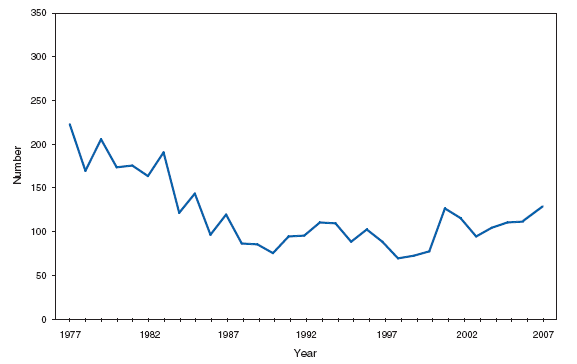
The incidence of brucellosis in the United States has been increasing slightly in recent years. Although brucellosis in U.S. cattle is in the final stages of eradication, the disease persists in feral swine, elk, and bison, increasing the risk of transmission to hunters while cleaning and dressing these animals. Outside of the United States, brucellosis remains endemic in a number of areas, including Mexico and the Mediterranean region, which poses a high risk of infection to travelers who consume unpasteurized milk products, including soft cheeses. Brucellosis. Number of reported cases --- United States and U.S. territories, 2007 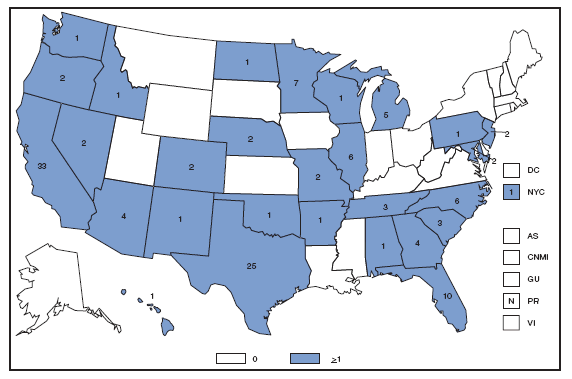
Reports of brucellosis cases are more frequent along the southern U.S. border, as the disease remains endemic in Mexico. Consumption of unpasteurized milk products, including soft cheeses from regions where brucellosis is common in cattle, sheep, and goats, presents a significant risk. Brucellosis caused by contact with infected feral swine while hunting has been documented in Florida and California. Chlamydia. Incidence* among women --- United States and U.S. territories, 2007 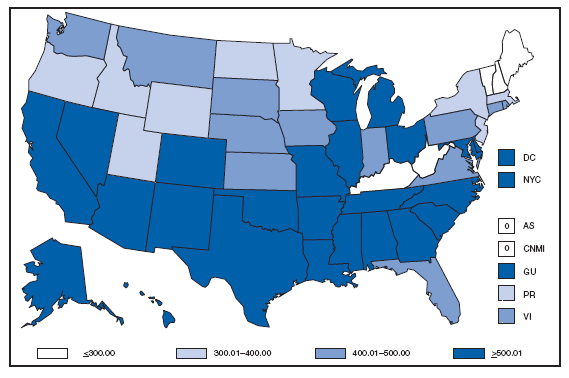
* Per 100,000 population. Chlamydia refers to genital infections caused by Chlamydia trachomatis. In 2007, the chlamydia rate among women in the U.S. and territories (Guam, Puerto Rico, and Virgin Islands) was 540.9 cases per 100,000 population. Cholera. Number of reported cases --- United States and U.S. territories, 2007 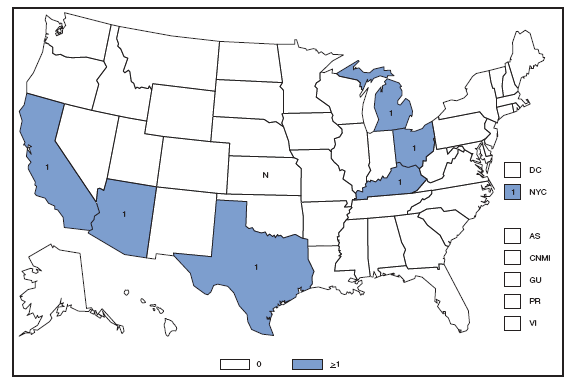
In 2007, approximately half of the infections of cholera were acquired during travel to Southeast Asia; the other half were acquired through the consumption of domestic seafood. Foreign travel and the consumption of contaminated domestic seafood remain important sources of cholera infection. Coccidioidomycosis. Number of reported cses --- United States* and U.S. territories, 2007 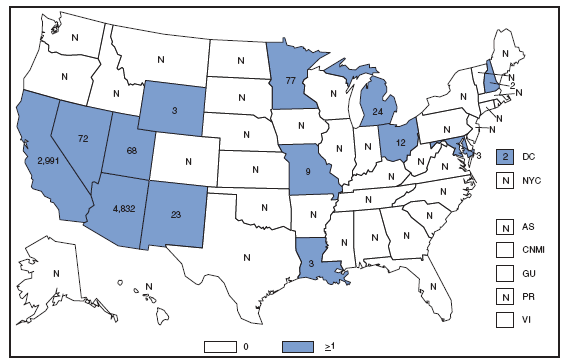
*In the United States, coccidioidomycosis is endemic to the southwestern States. However, cases have been reported in other states, usually among travelers returning from areas in which the disease is endemic. Reports of coccidioidomycosis cases decreased slightly nationwide in 2007, reflecting a decrease in the number of cases reported from California and Arizona. Cases reported from outside the disease-endemic states of Arizona, California, Nevada, New Mexico, Texas, and Utah likely represent exposure during travel to a disease-endemic area. Cryptosporidiosis. Incidence,* by year --- United States, 1996--2007 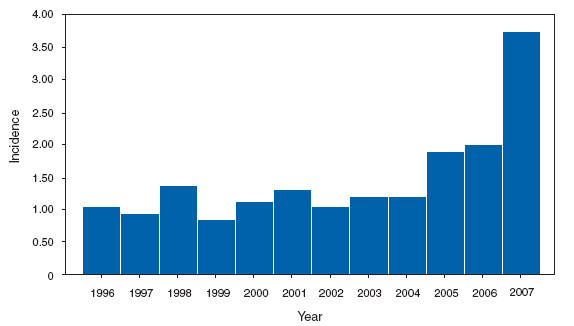
* Per 100,000 population. The increase in the incidence of cryptosporidiosis that began in 2005 accelerated in 2007. Whether this increase reflects changing diagnostic testing and reporting patterns or a real increase in cryptosporidiosis is unclear. Cryptosporidiosis. Incidence* --- United States and U.S. territories, 2007 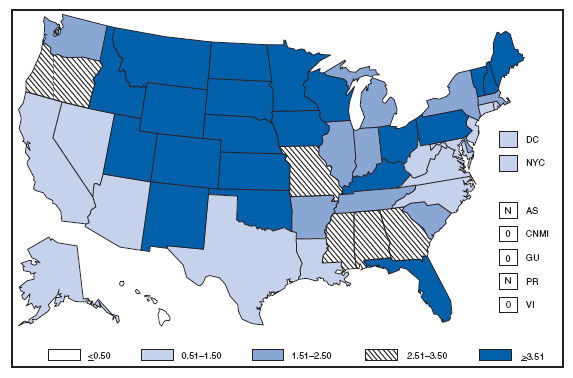
* Per 100,000 population. Cryptosporidiosis is widespread geographically in the United States, and differences in incidence between states might be affected by the capacity of states to detect and report cases. Although increased diagnosis and reporting has been noted previously in northern states, cryptosporidiosis has increased in nearly all states. Increased onset of illness occurs during summer, coinciding with increased use of recreational water. Diphtheria. Number of reported cases, by year --- United States, 1977--2007 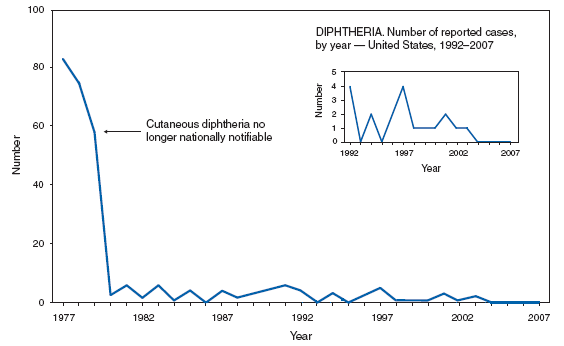
Since 2004, no case of respiratory diphtheria has been reported in the United States, and the national health objective for 2010 of zero cases has been maintained. DOMESTIC ARBOVIRAL DISEASES. Number* of reported cases of neuroinvasive disease, by year --- United States, 1998--2007 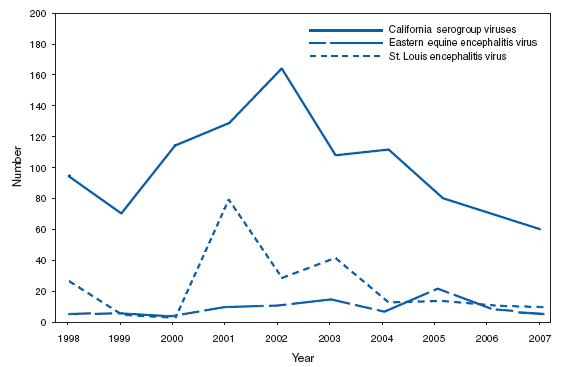
* Data from the Division of Vector-Borne Infectious Diseases, National Center for Zoonotic, Vector-Borne, and Enteric Diseases (ArboNET Surveillance). Only reported cases of neuroinvasive disease are shown. Arthropod-borne viruses (arboviruses) are transmitted primarily during the summer and fall in the United States, with the incidence of human disease peaking in the late summer. The most common arboviruses affecting humans in the United States are West Nile virus (WNV), La Crosse virus (LACV), eastern equine encephalitis virus (EEEV), and St. Louis encephalitis virus (SLEV). LACV is the most common California (CAL) serogroup virus in the United States. LACV causes neuroinvasive disease primarily among children. In 2007, CAL serogroup virus disease cases were reported from 10 states (Georgia, Illinois, Iowa, Minnesota, New York, North Carolina, Ohio, Tennessee, West Virginia, and Wisconsin). During 1998--2007, a median of 105 (range: 55--167) cases per year were reported in the United States. EEEV disease in humans is associated with high mortality rates (>20%) and severe neurologic sequelae. In 2007, EEEV cases were reported from two states (Alabama and New Hampshire). During 1998--2007, a median of seven (range: 3--21) cases per year were reported in the United States. Before the introduction of WNV to the United States, SLEV was the nation's leading cause of epidemic viral encephalitis. In 2007, SLEV cases were reported from five states (Arkansas, Louisiana, Missouri, Mississippi, and Nevada). During 1998--2007, a median of 11 (range: 2--79) cases per year were reported in the United States. DOMESTIC ARBOVIRAL DISEASES, WEST NILE. Incidence* of reported cases of neuroinvasive disease, by county --- United States, 2007 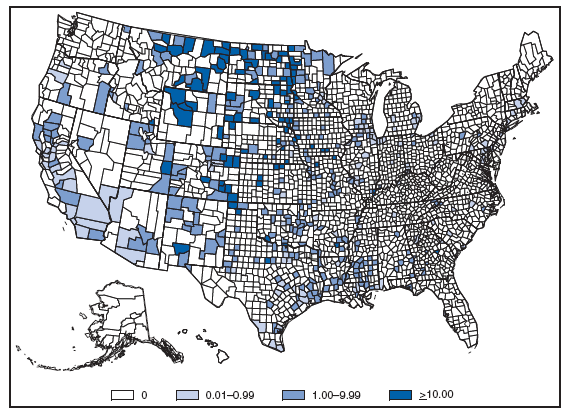
* Per 100,000 population. In 2007, the reported incidence of West Nile virus neuroinvasive disease in the United States was 0.4 per 100,000 population. This incidence is similar to that reported in 2004 (0.4), 2005 (0.4), and 2006 (0.5) but substantially lower than the reported incidence for 2002 (1.0) and 2003 (1.0). The highest incidence occurred primarily in the west-central United States. The five states with highest incidence were North Dakota (7.7), South Dakota (6.2), Wyoming (4.6), Montana (4.0), and Colorado (2.2). DOMESTIC ARBOVIRAL DISEASES, WEST NILE. Incidence* of reported cases of neuroinvasive disease, by age group --- United States, 2007 
* Per 100,000 population. In 2007, the median age of patients with West Nile virus neuroinvasive disease was 57 years (range: 1 month -- 97 years), with increasing incidence among older age groups. Ehrlichiosis, Human Granulocytic. Number of reported cases, by county --- United States, 2007 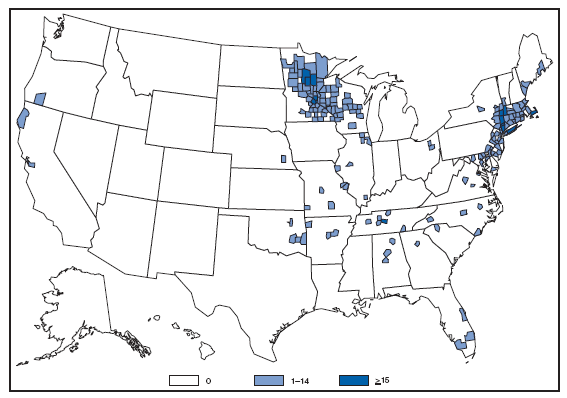
Human granulocytic ehrlichiosis (HGE), caused by Anaplasma phagocytophilum, is more correctly referred to as anaplasmosis as a result of recent taxonomic changes. Cases are reported primarily from the upper Midwest and coastal New England, reflecting both the range of the primary tick vector species (Ixodes scapularis) and the range of preferred animal hosts for tick feeding. Ehrlichiosis, Human Monocytic. Number of reported cases, by county --- United States, 2007 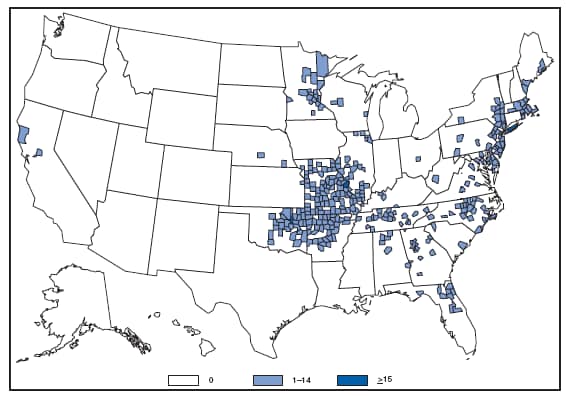
Human monocytic ehrlichiosis (HME), caused by Ehrlichia chaffeensis, is the most common type of ehrlichiosis. Cases are reported primarily in the lower Midwest and the Southeast, reflecting the range of the primary tick vector species (Amblyomma americanum). Ehrlichiosis, Human (other & unspecified). Number of reported cases, by county --- United States, 2007 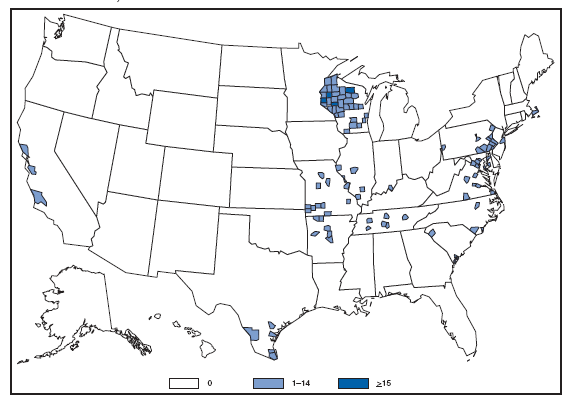
Cases of ehrlichiosis caused by other species (such as E. ewingii), or more commonly, cases for which the geographically expected species is not clearly differentiated by serologic testing, are reflected in this reporting category. Because Ehrlichia and Anaplasma infections might elicit cross-reactive antibody responses, some states might also use this category to report cases for which single, inappropriate diagnostic tests were run (e.g., physicians ordering only HME tests in a region where HGE would be expected to predominate). Giardiasis. Incidence* --- United States and U.S. territories, 2007 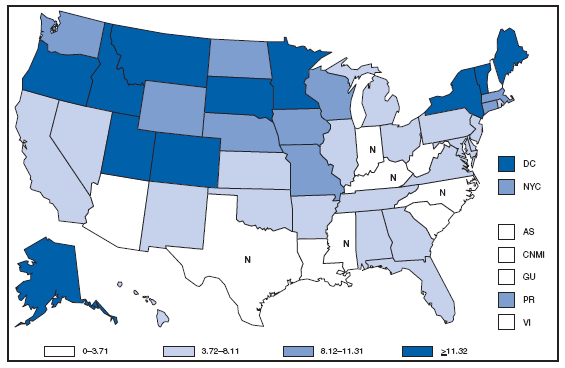
* Per 100,000 population. Giardiasis is widespread geographically in the United States, with increased reporting of giardiasis in northern states. However, because differences in giardiasis surveillance systems among states can affect the capability to detect cases, whether this finding is of true biologic significance or is the result of differences in case detection or reporting is difficult to determine. Gonorrhea. Incidence* --- United States and U.S. territories, 2007 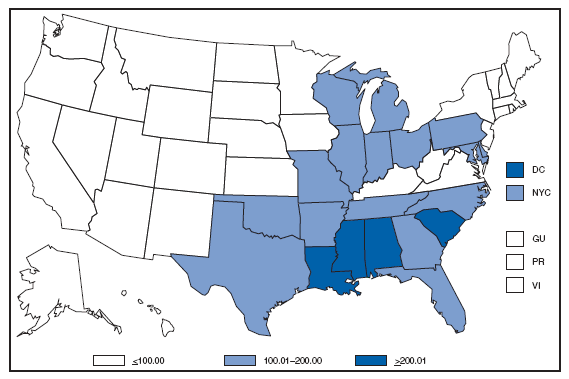
* Per 100,000 population. In 2007, the gonorrhea rate in the United States and U.S. territories (Guam, Puerto Rico, and Virgin Islands) was 117.4 cases per 100,000 population, a decrease from the rate in 2006. The Healthy People 2010 national objective is =19 cases per 100,000 population. Seven states (Idaho, Maine, Montana, New Hampshire, North Dakota, Vermont, and Wyoming) and Puerto Rico reported rates below the national objective. Gonorrhea. Incidence,* by sex --- United States, 1992--2007 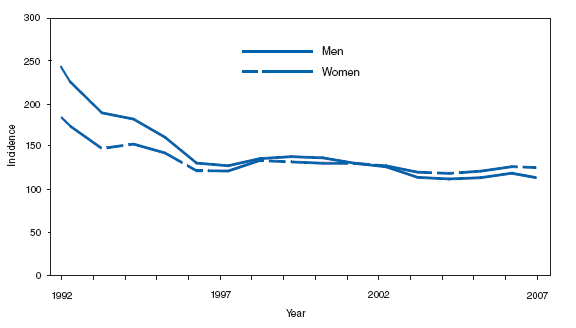
* Per 100,000 population. After a 74% decline in the rate of reported gonorrhea during 1975--1997, overall gonorrhea rates remained stable. In 2007, for the seventh year in a row, the gonorrhea rate among women was slightly higher than the rate among men. Gonorrhea. Incidence,* by race/ethnicity --- United States, 1992--2007 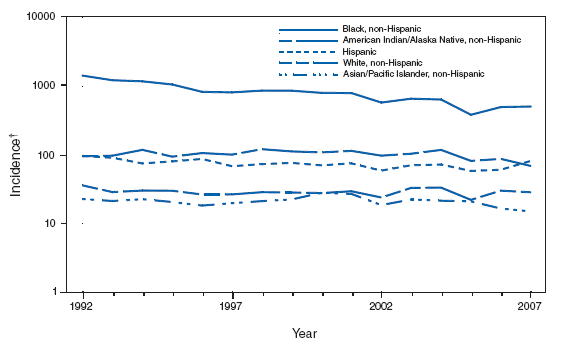
* Per 100,000 population. † Y-axis is log scale. Gonorrhea incidence among blacks decreased considerably during the 1990s but continues to be the highest among all races/ethnicities. In 2007, incidence among non-Hispanic blacks was approximately 19 times greater than that for non-Hispanic whites. Haemophilus influenzae, Invasive Disease. Incidence, * by age group --- United States, 1994--2007 
* Per 100,000 population. Substantial reductions in the incidence of Haemophilus influenzae serotype b (Hib) disease have been achieved through universal Hib vaccination. Before introduction of conjugate vaccines in 1987, the incidence of invasive Hib disease among children aged <5 years was estimated to be 100 cases per 100,000 population. To monitor the epidemiology of Hib invasive disease and to detect the emergence of invasive non-Hib, serotyping of all Hi isolates in children aged <5 years and thorough and timely investigation of all cases of Hib disease are essential. Hemolytic Uremic Syndrome, Postdiarrheal. Number of reported cases --- United States and U.S. territories, 2007 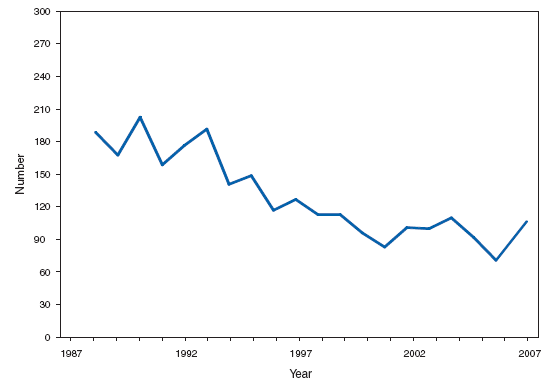
In the United States, the majority of cases of postdiarrheal hemolytic uremic syndrome (HUS) are caused by infection with Escherichia coli O157:H7. Infection with other serotypes of Shiga toxin-producing E. coli can cause HUS. During 2007, as usual, the majority of reported cases occurred among children aged 1--4 years. HUS has been a nationally notifiable disease since 1995. Hansen Disease (Leprosy). Number of reported cases, by year --- United States, 1988--2007 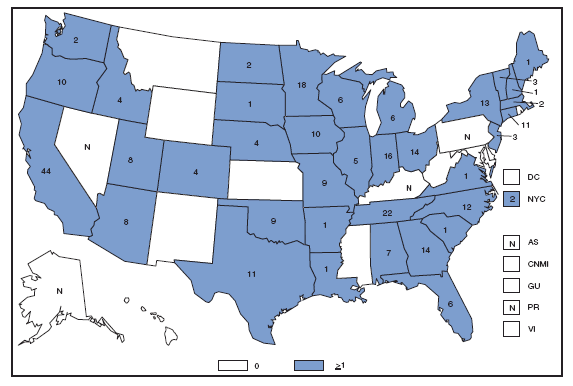
The number of cases of Hansen disease reported per year gradually declined during 1988--2006. Reported cases in 2007 increased 49% from 2006, which might reflect efforts CDC has made to improve reporting. Hepatitis, Viral. Incidence,* by year --- United States, 1977--2007 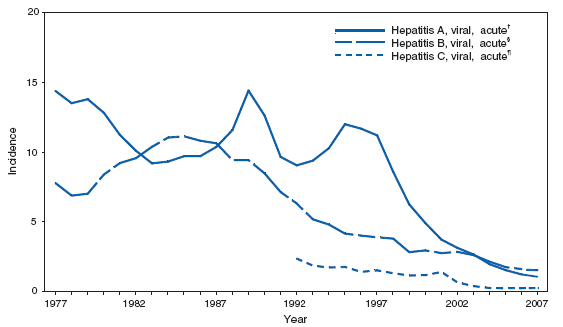
* Per 100,000 population. † Hepatitis A vaccine was first licensed in 1995. § Hepatitis B vaccine was first licensed in June 1982. ¶ An anti-hepatitis C virus (HCV) antibody test first became available in May 1990. Hepatitis A incidence continues to decline and in 2007 was the lowest ever recorded. This reduction in incidence is attributable at least in part to routine vaccination of children. Hepatitis A incidence has declined 90% since the last nationwide outbreak in 1995. Routine hepatitis B vaccination of infants has reduced rates >95% in children. Rates have also declined among adults, but a substantial proportion of cases continue to occur among adults with high-risk behaviors. Incidence of acute hepatitis C has declined approximately 90% since 1992; however, a substantial burden of disease as a result of chronic HCV infection remains. Hepatitis A. Incidence* by county --- United States, 2007 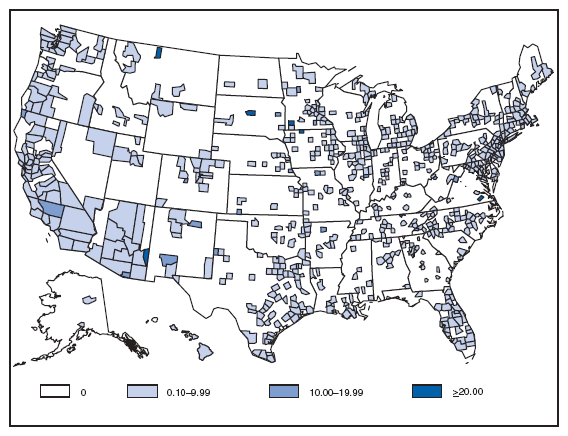
* Per 100,000 population. In 1999, routine hepatitis A vaccination was recommended for children living in 11 states with consistently elevated rates of disease. Since then, rates of infection with hepatitis A virus (HAV) have declined in all regions, with the greatest decline occurring in western states. HAV infection rates are now the lowest ever reported and similar in all regions. As of 2005, hepatitis A vaccine is now recommended for children in all states. Influenza-Associated Pediatric Mortality. Incidence* --- United States and U.S. territories, 2007 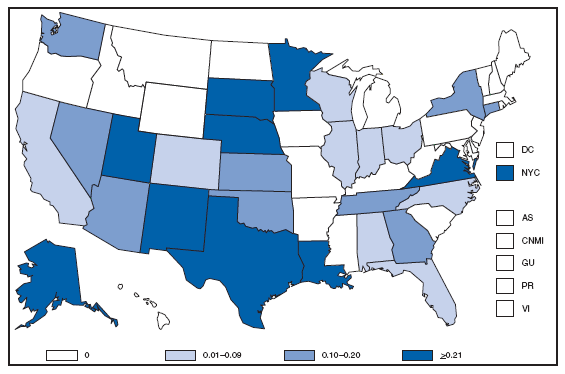
* Per 100,000 population. During 2007, a total of 77 influenza-associated pediatric deaths were reported to CDC by 27 states, New York City, and Chicago, for an overall incidence rate in the United States of 0.10 deaths per 100,000 children aged <18 years. This is similar to rates estimated through mathematical modeling. State-to-state variation in rates was small and likely reflected the rarity of the event and small population size rather than true differences in disease. Legionellosis. Incidence, * by year --- United States, 1992--2007 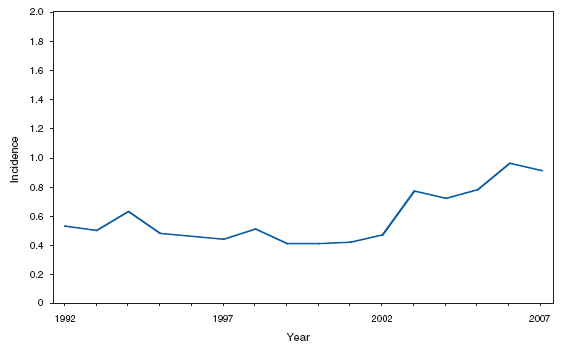
* Per 100,000 population. Legionellosis incidence declined slightly in 2007 compared with 2006. Factors contributing to an overall increase in recent years might include a true increase in disease transmission, greater use of diagnostic testing, and increased reporting. Listeriosis. Incidence* --- United States and U.S. territories, 2007 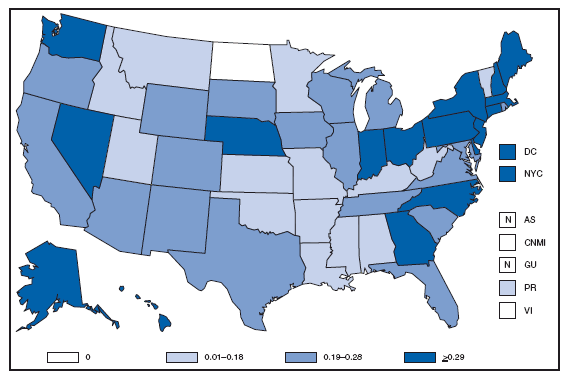
* Per 100,000 population. Listeriosis is primarily foodborne and occurs most frequently among persons who are older, pregnant, or immunocompromised. Recent outbreaks have been associated with deli meats and unpasteurized cheeses. Lyme Disease. Number of reported cases, by county --- United States, 2007 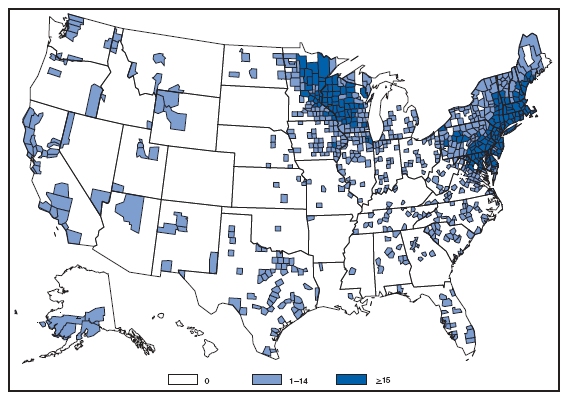
Over 95% of Lyme disease cases are reported from states in the northeastern and upper midwestern United States. A rash that can be confused with early Lyme disease sometimes occurs following bites of the lone star tick (Amblyomma americanum). These ticks, which do not transmit the Lyme disease bacterium, are common human-biting ticks in southern and southeastern United States. Malaria. Incidence, * by year --- United States, 1993--2007 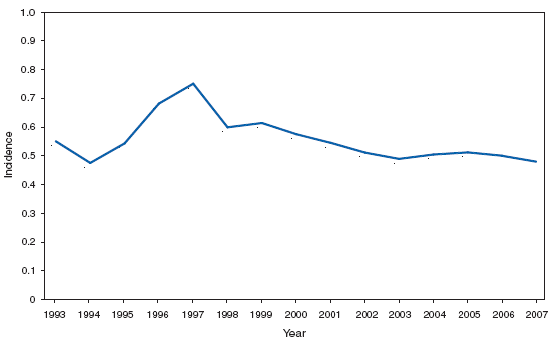
* Per 100,000 population. The number of reported cases of malaria in the United States has remained relatively stable for the preceding 5 years. Nearly all of these infections occur in persons who traveled recently to a malaria-endemic country. Measles. Incidence, * by year --- United States, 1972--2007 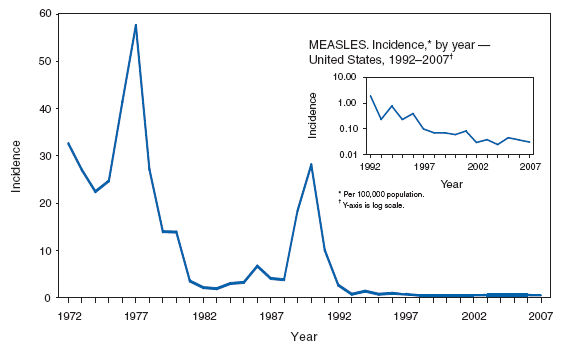
* Per 100,000 population. Measles vaccine was licensed in 1963. Evidence suggests that measles is no longer endemic in the United States. Meningococcal disease. Incidence, * by year --- United States, 1977--2007 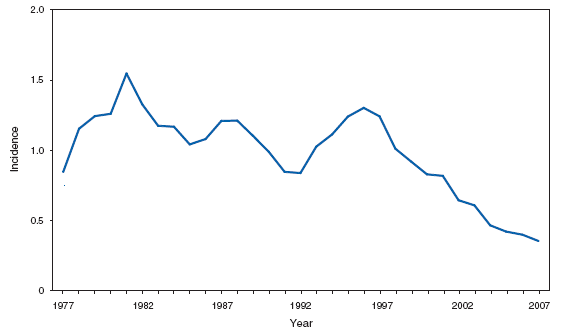
* Per 100,000 population. Meningococcal disease incidence is currently at a historic low but continues to cause substantial morbidity and mortality in the United States. The highest incidence of meningococcal disease occurs among infants, with a second peak occurring during late adolescence. In 2005, a tetravalent (A, C, Y, W-135) meningococcal conjugate vaccine was licensed and recommended for adolescents and others at increased risk for disease. Over time, the new vaccine is expected to have a substantial impact on the burden of meningococcal disease in the United States. Mumps. Incidence, * by year --- United states, 1982--2007 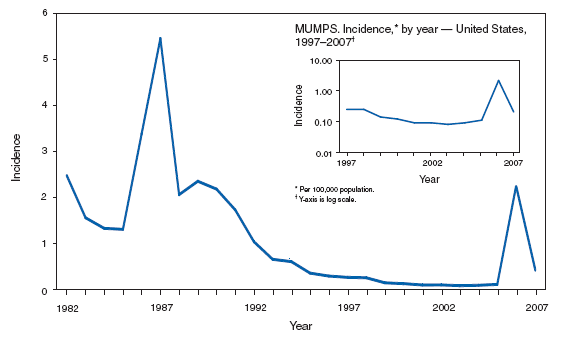
* Per 100,000 population. Mumps vaccine was licensed in 1967. The widespread use of a second dose of mumps vaccine in 1990 was followed by historically low morbidity until 2006, when the United States experienced the largest mumps outbreak in 2 decades. The 2006 outbreak of >6,000 cases affected primarily college students aged 18--24 years in the Midwest. As a result, the Advisory Committee on Immunization Practices (ACIP) updated its vaccination recommendations, and the Council of State and Territorial Epidemiologists (CSTE) updated its case definition. PERTUSSIS. Incidence,* by year --- United States, 1977--2007 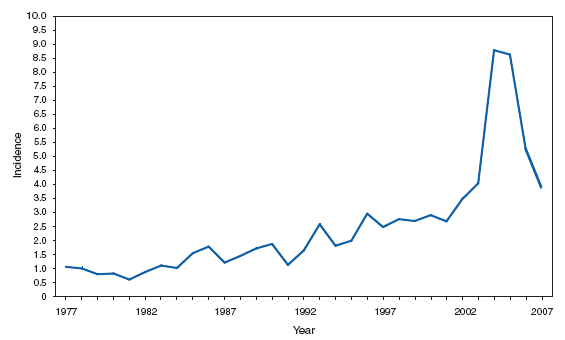
* Per 100,000 population. In 2007, incidence of reported pertussis dropped sharply from the peak in 2004 but remains higher than in the 1990s. PERTUSSIS. Number of reported cases,* by age group --- United States, 2007 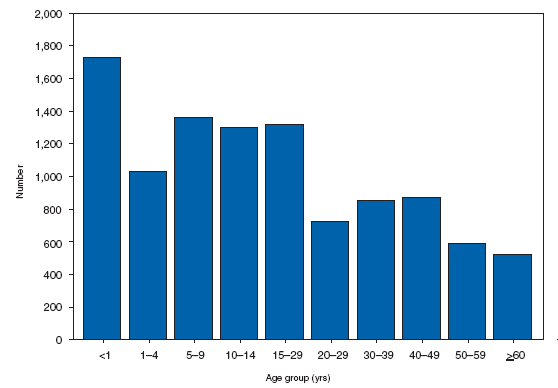
* Of 10,454 cases, age was reported unknown for 212 persons. Infants, especially those who are undervaccinated, are at increased risk for complicated infections and death from pertussis. Immunity to pertussis wanes 5--10 years after completion of childhood vaccination. A second peak in the number of reported cases is observed in adolescents. Q Fever. Number of reported cases --- United States and U.S. territories, 2007 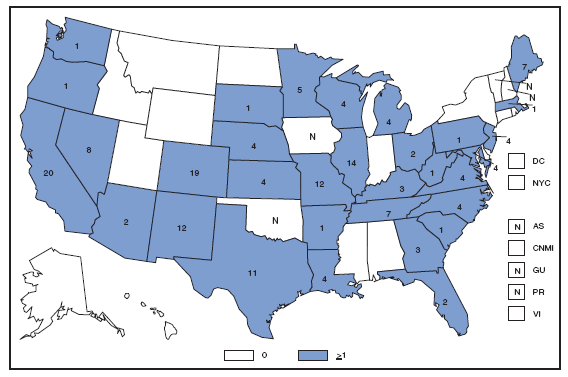
Q fever, caused by Coxiella burnetii, is reported throughout the United States. Human cases occur as a result of human interaction with livestock, especially sheep, goats, and cattle. Although relatively few human cases are reported annually, the disease is believed to be substantially underreported because of its nonspecific presentation and the subsequent failure to suspect infection and request appropriate diagnostic tests. Rabies, Animal. Number of reported cases among wild and domestic animals, * by year --- United States and Puerto Rico, 1977--2007 
* Data from the National Center for Zoonotic, Vector-Borne, and Enteric Diseases. † In thousands. Periods of resurgence and decline of rabies incidence result primarily from cyclic reemergence. The recent increase of 1.7% in the number of reported cases from 2006 follows the current increasing trend. Although numeric increases are subject to surveillance bias, the proportion of positive cases among tested animals remained relatively stable during 2007. Recent increases in the number of reported cases of rabies in bats have led to this order of mammals becoming the second-most-reported group with rabies after raccoons. Rocky Mountain Spotted Fever. Number of reported cases, by county --- United States, 2007 
Rocky Mountain spotted fever caused by Rickettsia rickettsii, is reported throughout much of the United States, reflecting the widespread ranges of the primary tick vectors responsible for transmission (primarily Dermacentor variabilis in the East and Dermacentor andersonii in the West, but also Rhipicephalus sanguineus in some newly recognized focal areas). Rubella. Incidence, * by year --- United States, 1977--2007 
* Per 100,000 population. Rubella vaccine was licensed in 1969. Evidence suggests that rubella is no longer endemic in the United States. Salmonellosis and Shigellosis. Number* of reported cases, by year --- United States, 1977--2007 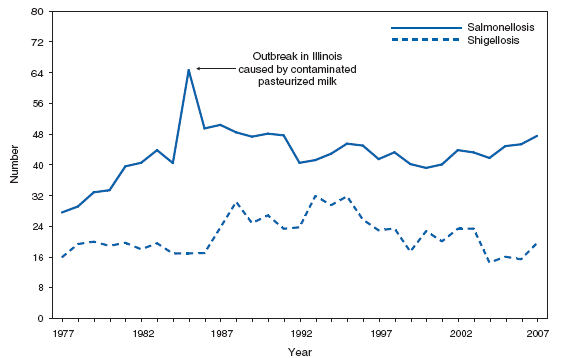
* In thousands. Rates of salmonellosis have remained relatively stable of the past decade. Serotypes Typhimurium, Enteritidis, and Newport are the most commonly reported Salmonella serotypes Shiga toxin-producing Escherichia coli (STEC). Number of reported cases --- United States and U.S. territories, 2007 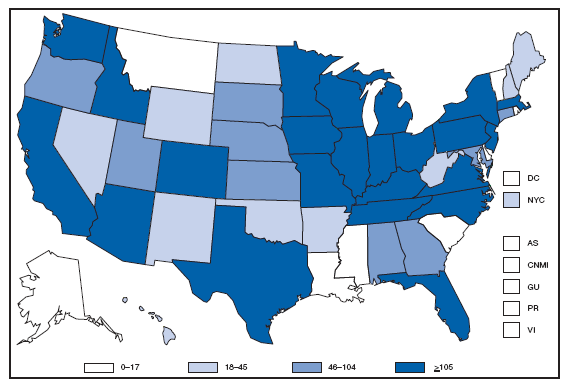
Escherichia coli O157:H7 is the serotype of Shiga toxin-producing E. coli (STEC) isolated most commonly from ill persons. Other serotypes of E. coli also produce Shiga toxin and can cause diarrhea and hemolytic uremic syndrome. E. coli O157:H7 has been nationally notifiable since 1994. STEC that cause human illness sometimes are called enterohemorrhagic E. coli (EHEC). In 2001, all serotypes of EHEC were made nationally notifiable. In 2006, the National Notifiable Diseases Surveillance System designation was changed by the Council of State and Territorial Epidemiologists from enterohemorrhagic E. coli (EHEC) to STEC, and reporting of serotypes to CDC was strongly encouraged. Streptococcal Disease, invasive, Group A. Number of reported cases --- United States and U.S. territories, 2007 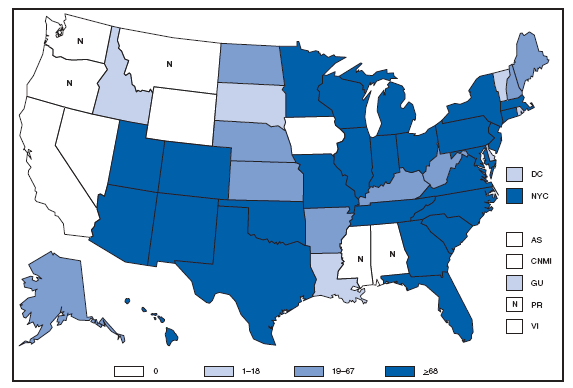
Completeness of reporting of invasive group A streptococcal disease to the National Notifiable Diseases Surveillance System (NNDSS) is unknown. In 2007, the U.S. incidence rate of disease from NNDSS was 1.9 cases per 100,000. The NNDSS incidence rate excludes data from six states where this disease was not reportable (Alabama, Mississippi, Montana, California, Oregon, and Washington). In 2007, the estimated rate of disease from active, laboratory-based surveillance conducted in 10 U.S. sites was 3.8 cases per 100,000 population. Syphilis, Congenital. Incidence* among infants aged <1 year --- United States, 1977--2007 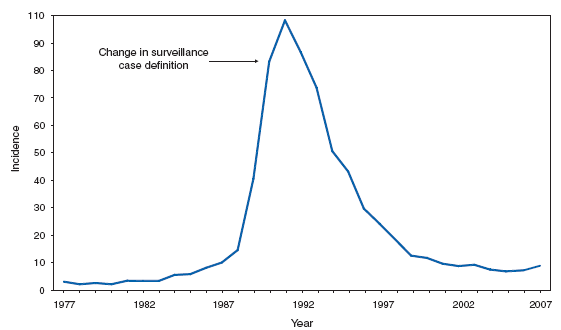
* Per 100,000 live births. Following a decline in the incidence of congenital syphilis since 1991, overall congenital syphilis rates increased slightly during 2006--2007, from 9.1 to 10.5 cases per 100,000 live births. Syphilis, Primary and Secondary. Incidence* --- United States and U.S. territories, 2007 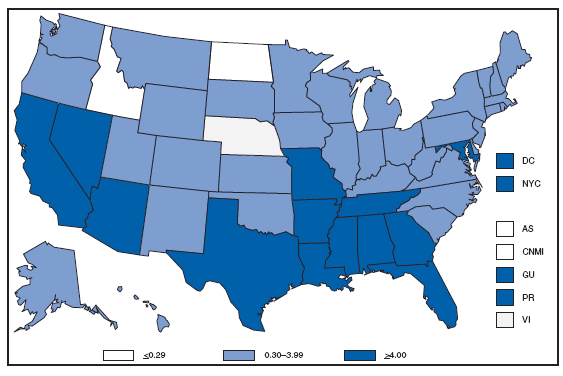
* Per 100,000 population. In 2007, the primary and secondary syphilis rate in the United States and territories (Guam, Puerto Rico, and Virgin Islands) was 3.8 cases per 100,000 population, which is above the Healthy People 2010 objective of 0.2 cases per 100,000 population per year. Three states (Idaho, Nebraska, and North Dakota) and the Virgin Islands reported rates at or below the national objective. Syphilis, Primary and Secondary. Incidence*, by sex --- United States, 1992--2007 
* Per 100,000 population. During 2006--2007, the incidence of primary and secondary syphilis in the United States increased from 3.3 to 3.8 cases (women: from 1.0 to 1.1; men: from 5.6 to 6.6) per 100,000 population. Syphilis, Primary and Secondary. Incidence,* by race/ethnicity --- United States, 1992--2007 
* Per 100,000 population. † Y-axis is log scale During 2006-2007, incidence of primary and secondary syphilis increased among all races/ethnicities except Asian/Pacific Islanders. Incidence per 100,000 population increased from 11.2 to 14.0 among non-Hispanic blacks, from 3.5 to 4.3 among Hispanics, from 3.2 to 3.4 among American Indian/Alaska Natives, and from 1.9 to 2.0 among non-Hispanic whites; incidence remained the same among Asian/Pacific Islanders at 1.2. Trichinellosis. Number of reported cases, by year --- United States, 1977--2007 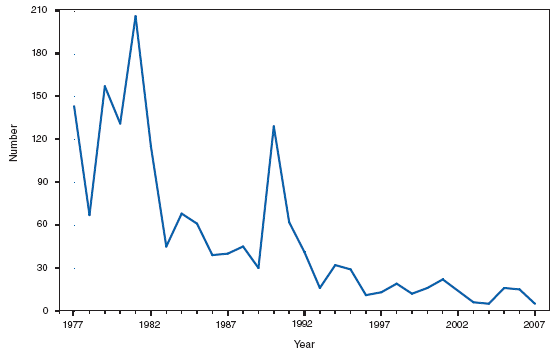
In 2007, five cases of trichinellosis were reported to CDC. Consumption of raw/undercooked bear meat was implicated in three cases, and for two of these cases, a meal of undercooked store-bought pork and bear sausage was shared. The remaining two persons had a history of consuming only store-bought pork and beef, respectively. Although improved methods of swine husbandry over the past several decades have made pork-associated cases of trichinellosis in the United States rare, consumption of wild game meat continues to be the most commonly identified risk factor for trichinellosis. Tuberculosis. Incidence* --- United States and U.S. territories, 2007 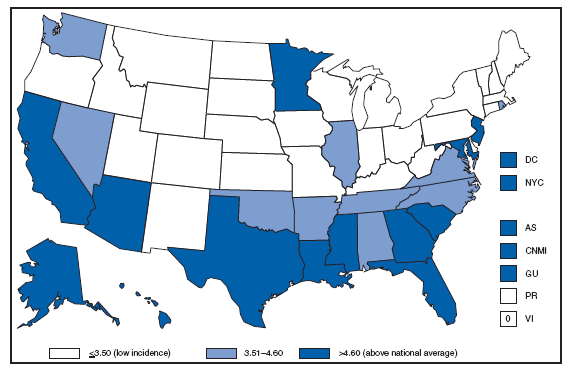
* Per 100,000 population. Fifteen states or reporting areas reported incidence rates above the national average in 2007. U.S.-affiliated areas are counted as separate nations and as such are not included in national totals. Tuberculosis. Number of reported cases among U.S.-born and foreign-born persons,* by year --- United States, 1997--2007 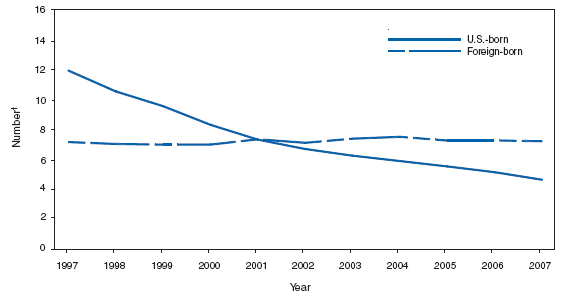
* For 56 cases, origin of patients was unknown † In thousands The proportion of total tuberculosis cases occurring in the foreign-born continues to rise. Tuberculosis. Incidence,* by race/ethnicity --- United States, 1997--2007 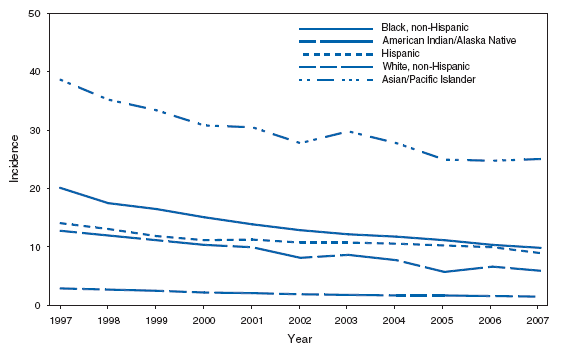
* Per 100,000 population. Rates for all race-ethnicities continued their downward trend in 2007 except among Asian/Pacific Islanders (APIs). Self-reported Native Hawaiians, a small portion of the API rate, experienced a 75% increase in 2007. Tularemia. Number of reported cases --- United States and U.S. territories, 2007 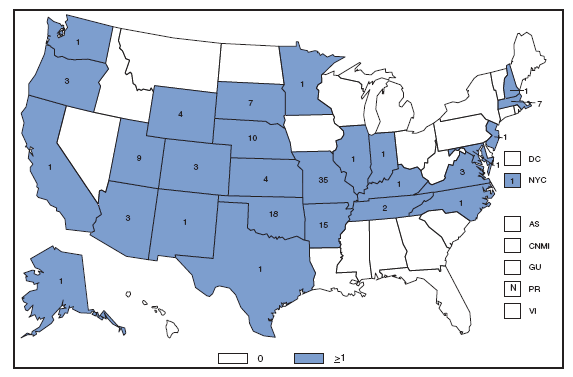
Historically, tularemia has been reported from all states except Hawaii. To define the geographic distribution of Francisella tularensis subspecies, CDC requests that isolates be forwarded to the CDC laboratory in Fort Collins, Colorado. Typhoid fever. Number of reported cases, by year --- United States, 1977--2007 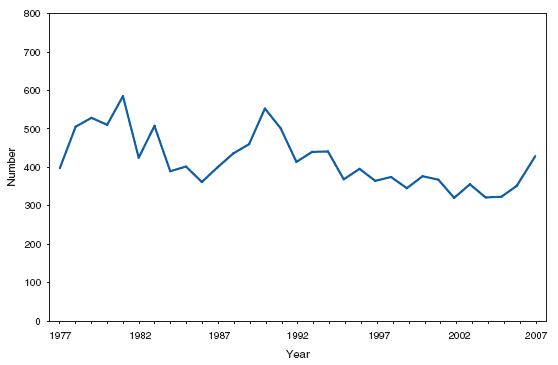
Typhoid fever in the United States is primarily a disease of travelers, for whom vaccination against typhoid fever is recommended. Emerging resistance to fluoroquinolone antimicrobial agents has complicated the clinical management of cases of typhoid and paratyphoid fever. Varicella (ChickenPox). Number of reported cases --- Illinois, Michigan, Texas, and West Virginia*, 1991--2007 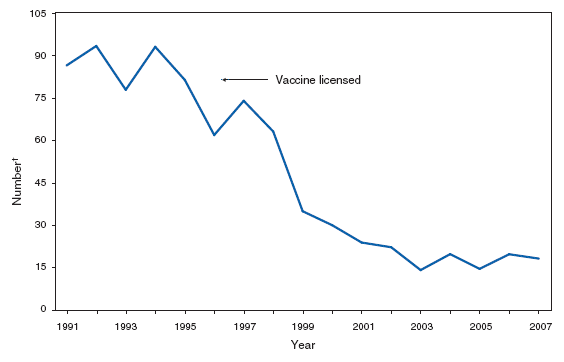
* Source: CDC. National Center for Immunization and Respiratory Diseases. † In thousands. In four states (Michigan, Illinois, Texas, and West Virginia), the number of cases reported in 2007 was 20% lower than in 2006 and 81% less than the number reported during the prevaccine years 1993--1995. Vibriosis. Number of reported cases --- United States and U.S. territories, 2007 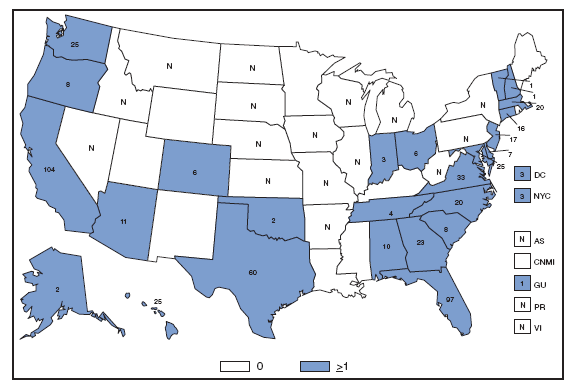
Vibriosis infections caused by noncholera Vibrio organisms became nationally notifiable in January 2007. Infections are acquired through consumption of contaminated seafood, particularly oysters, or by contact of broken skin to salt water, containing Vibrio organisms. Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. PART 3Historical Summaries of Notifiable Diseases in the United States, 1976--2007Abbreviations and Symbols Used in TablesNA Data not available. --- No reported cases. Notes: Rates <0.01 after rounding are listed as 0. Data in the MMWR Summary of Notifiable Diseases --- United States, 2007 might not match data in other CDC surveillance reports because of differences in the timing of reports, the source of the data, and the use of different case definitions.
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Selected ReadingGeneralOverhage JM, Grannis S, MdDonald CJ. A comparison of the completeness and timeliness of automated electronic laboratory reporting and spontaneous reporting of notifiable conditions. Am J Public Health 2008; Am J Public Health. 2008;98:344--50. Adekoya N. Nationally notifable disease surveillance (NNDSS) and the Healthy People 2010 objectives. The eJournal of the South Carolina Medical Association 2005;101:e68--72. Available at http://www.scmanet.org/Downloads/e-Journal/SCMA_eJournal_March05.pdf. Baker MG, Fidler DP. Global public health surveillance under new international health regulations. Emerg Infect Dis 2006;12:1058--65. Bayer R, Fairchild AL. Public health: surveillance and privacy. Science 2000;290:1898--9. CDC. Racial disparities in nationally notifiable diseases---United States, 2002. MMWR 2005;54:9--11. CDC. Case definitions for infectious conditions under public health surveillance. MMWR 1997;46(No. RR-10). Additional information available at http://www.cdc.gov/epo/dphsi/casedef/index.htm. CDC. Framework for program evaluation in public health. MMWR 1999;48(No. RR-11). CDC. Manual of procedures for the reporting of nationally notifiable diseases to CDC. Atlanta, GA: US Department of Health and Human Services, Public Health Service, CDC; 1995. CDC. Manual for the surveillance of vaccine-preventable diseases. 3rd ed. Atlanta, GA: US Department of Health and Human Services, Public Health Service, CDC; 2002. Available at http://www.cdc.gov/nip/publications/surv-manual. CDC. National Electronic Disease Surveillance System (NEDSS): a standards-based approach to connect public health and clinical medicine. J Public Health Management and Practice 2001;7:43--50. CDC. Public Health Information Network (PHIN): overview. Atlanta, GA: US Department of Health and Human Services, CDC; 2006. Available at http://www.cdc.gov/phin/overview.html. CDC. Sexually transmitted disease surveillance, 2007. Atlanta, GA: US Department of Health and Human Services, CDC; 2008. Available at http://www.cdc.gov/std/stats. CDC. Sexually transmitted diseases treatment guidelines, 2006. MMWR 2006;55(No. RR-11). Chang M-H, Glynn MK, Groseclose SL. Endemic, notifiable bioterrorism-related diseases, United States, 1992--1999. Emerg Infect Dis 2003;9:556--64. Chin JE, ed. Control of communicable diseases manual. 17th ed. Washington, DC: American Public Health Association; 2000. Doyle TJ, Glynn MK, Groseclose SL. Completeness of notifiable infectious disease reporting in the United States: an analytical literature review. Am J Epidemiol 2002;155:866--74. Effler P, Ching-Lee M, Bogard A, Ieong M-C, Nekomoto T, Jernigan D. Statewide system of electronic notifiable disease reporting from clinical laboratories: comparing automated reporting with conventional methods. JAMA 1999;282:1845--50. Freimuth V, Linnan HW, Potter P. Communicating the threat of emerging infections to the public. Emerg Infect Dis 2000;6:337--47. German R. Sensitivity and predictive value positive measurements for public health surveillance systems. Epidemiology 2000;11:720--7. Government Accountability Office. Emerging infectious diseases: review of state and federal disease surveillance efforts. Washington, DC: Government Accountability Office; 2004. GAO-04-877. Available at http://www.gao.gov/new.items/d04877.pdf. Hopkins RS. Design and operation of state and local infectious disease surveillance systems. J Public Health Manag Pract 2005;11:184--90. Jajosky RA, Groseclose SL. Evaluation of reporting timeliness of public health surveillance systems for infectious diseases. BMC Public Health 2004;4:29. Koo D, Caldwell B. The role of providers and health plans in infectious disease surveillance. Eff Clin Pract 1999;2:247--52. Available at http://www.acponline.org/journals/ecp/sepoct99/koo.htm. Koo D, Wetterhall S. History and current status of the National Notifiable Diseases Surveillance System. J Public Health Manag Pract 1996;2:4--10. Krause G, Brodhun B, Altmann D, Claus H, Benzler J. Reliability of case definitions for public health surveillance assessed by Round-Robin test methodology. BMC Public Health 2006;6:129. Lazarus R, Klompas M, Campion F, et al. Electronic support for public health: validated case finding and reporting for notifiable diseases using electronic medical data. J Am Med Inform Assoc 2009;16(1):18--24. Lin SS, Kelsey JL. Use of race and ethnicity in epidemiologic research: concepts, methodological issues, and suggestions for research. Epidemiol Rev 2000;22:187--202. Martin SM, Bean NH. Data management issues for emerging diseases and new tools for managing surveillance and laboratory data. Emerg Infect Dis 1995;1:124--8. McNabb S, Chungong S, Ryan M, et al. Conceptual framework of public health surveillance and action and its application in health sector reform. BMC Public Health 2002;2:2. McNabb S, Surdo A, Redmond A, et al. Applying a new conceptual framework to evaluate tuberculosis surveillance and action performance and measure the costs, Hillsborough County, Florida, 2002. Ann Epidemiol 2004;14:640--5. Niskar AS, Koo D. Differences in notifiable infectious disease morbidity among adult women---United States, 1992--1994. J Womens Health 1998;7:451--8. Panackal AA, M’ikanatha NM, Tsui FC, et al. Automatic electronic laboratory-based reporting of notifiable infectious diseases at a large health system. Emerg Infect Dis 2002;8:685--91. Pinner RW, Koo D, Berkelman RL. Surveillance of infectious diseases. In: Lederberg J, Alexander M, Bloom RB, eds. Encyclopedia of microbiology. 2nd ed. San Diego, CA: Academic Press; 2000. Pinner RW, Jernigan DB, Sutliff SM. Electronic laboratory-based reporting for public health. Mil Med 2000;165(Suppl 2):20--4. Roush S, Birkhead G, Koo D, Cobb A, Fleming D. Mandatory reporting of diseases and conditions by health care professionals and laboratories. JAMA 1999;282:164--70. Roush S, Murphy T. Historical comparisons of morbidity and mortality for vaccine-preventable diseases in the United States. JAMA 2007;298:2155--63. Silk, BJ, Berkelman RL. A review of strategies for enhancing the completeness of notifiable disease reporting. J Public Health Manag Pract 2005;11:191--200. Teutsch SM, Churchill RE, eds. Principles and practice of public health surveillance. 2nd ed. New York, NY: Oxford University Press; 2000. Thacker SB, Choi K, Brachman PS. The surveillance of infectious diseases. JAMA 1983;249:1181--5. AIDSCDC. HIV/AIDS surveillance report, 2007. Atlanta, GA: US Department of Health and Human Services, CDC, Vol. 19; 2009. Available at http://www.cdc.gov/hiv/topics/surveillance/resources/reports. AnthraxBlackburn JK, McNyset KM, Curtis A, Hugh-Jones ME. Modeling the geographic distribution of Bacillus anthracis, the causative agent of anthrax disease, for the contiguous United States using predictive ecological niche modeling. Am J Trop Med Hyg 2007;77:1103--10. Inglesby TV, O’Toole T, Henderson DA, et al. Anthrax as a biological weapon, 2002: updated recommendations for management. JAMA 2002;287:2236--52. Stern EJ, Uhde KB, Shadomy SV, Messonnier N. Conference report on public health and clinical guidelines for anthrax. Emerg Infect Dis 2008;14. Available at http://www.cdc.gov/EID/content/14/4/07-0969.htm. BotulismAngulo FJ, St. Louis ME. Botulism. In: Evans AS, Brachman PS, eds. Bacterial infections of humans. New York, NY: Plenum; 1998:131--53. CDC. Infant botulism---New York City, 2001--2002. MMWR 2003;52:21--4. Shapiro RL, Hatheway C, Becher J, Swerdlow DL. Botulism surveillance and emergency response: a public health strategy for a global challenge. JAMA 1997;278:433--5. Shapiro RL, Hatheway C, Swerdlow DL. Botulism in the United States: a clinical and epidemiologic review. Ann Intern Med 1998;129:221--8. Sobel J, Tucker N, McLaughlin J, Maslanka S. Foodborne botulism in the United States, 1999--2000. Emerg Infect Dis 2004;10:1606--12. Sobel J. Botulism. Clin Infect Dis 2005;41:1167--73. BrucellosisCDC. Brucellosis (Brucella melitensis, abortus, suis, and canis). Atlanta, GA: US Department of Health and Human Services, CDC; 2005. Available at http://www.cdc.gov/ncidod/dbmd/diseaseinfo/brucellosis_g.htm. CDC. Brucellosis case definition. Atlanta, GA: US Department of Health and Human Services, CDC; 2001. Available at http://www.bt.cdc.gov/Agent/Brucellosis/CaseDef.asp. CDC. Human exposure to Brucella abortus strain RB51---Kansas, 1997. MMWR 1998;47:172--5. Stevens, MG, Olsen SC, Palmer MV, Cheville NF. US Department of Agriculture, Agricultural Research Service National Animal Disease Center, Iowa State University. Brucella abortus strain RB51: a new brucellosis vaccine for cattle. Compendium 1997;19:766--74. Yagupsky P, Baron EJ. Laboratory exposures to Brucellae and implications for bioterrorism. Emerg Infect Dis 2005;11:1180--5. Chomel BB, DeBess EE, Mangiamele DM, et al. Changing trends in the epidemiology of human brucellosis in California from 1973 to 1992: a shift toward foodborne transmission. J Infect Dis 1994;170:1216--23. ChancroidDiCarlo RP, Armentor BS, Martin DH. Chancroid epidemiology in New Orleans men. J Infect Dis 1995;172:446--52. Mertz KJ, Weiss JB, Webb RM, et al. An investigation of genital ulcers in Jackson, Mississippi, with use of a multiplex polymerase chain reaction assay: high prevalence of chancroid and human immunodeficiency virus infection. J Infect Dis 1998;178:1060--6. Mertz KJ, Trees D, Levine WC, et al. Etiology of genital ulcers and prevalence of human immunodeficiency virus coinfection in 10 US cities. The Genital Ulcer Disease Surveillance Group. J Infect Dis 1998;178:1795 Chlamydia trachomatis, Genital InfectionCDC. Sexually transmitted disease surveillance, 2007 Supplement, Chlamydia Prevalence Monitoring Project Annual Report 2007. Atlanta, GA: U.S. Department of Health and Human Services, CDC; 2009. Available at http://www.cdc.gov/std/chlamydia2007. Datta SP, Sternberg M, Johnson RE, et al. Gonnorhea and chlamydia in the United States among persons 14 to 39 years of age, 1999 to 2002. Ann Intern Med 2007;147:89--96. Miller WC, Ford CA, Handcock MS, et al. Prevalence of chlamydial and gonococcal infections among young adults in the United States. JAMA 2004;291:2229--36. Satterwhite CL, Joesoef MR, Datta SD, Weinstock H. Estimates of Chlamydia trachomatis infections among men: United States. Sexually Trans Dis 2007;35: S3--7. CholeraSteinberg EB, Greene KD, Bopp CA, Cameron DN, Wells JG, Mintz ED. Cholera in the United States, 1995--2000: trends at the end of the millennium. J Infect Dis 2001;184:799--802. World Health Organization. Cholera, 2007. Wkly Epidemiol Rec 2008;81:269--84. Gaffga NH, Tauxe RV, Mintz ED. Cholera: a new homeland in Africa. Am J Trop Med Hyg 2007;77:705--13. Tobin-D’Angelo M, Smith AR, Bulens SN, et al. Severe diarrhea caused by cholera toxin--producing Vibrio cholerae serogroup O75 infections acquired in the southeastern United States. Clin Infect Dis 2008;47:1035--40. CoccidioidomycosisChang DC, Anderson S, Wannemuehler K, et al. Infrequent testing for coccidioidomycosis among patients with community-acquired pneumonia: implications for public health in an endemic area. Emerg Infect Dis 2008;14:1053--9. Valdivia L, Nix D, Wright M, et al. Coccidioidomycosis as a common cause of community-acquired pneumonia. Emerg Infect Dis 2006;12: 958--62. Lee R, Crum-Cianflone N, Increasing incidence and severity of coccidioidomycosis at a naval air station. Mil Med 2008;173:769--75. CryptosporidiosisRoy SL, DeLong SM, Stenzel S, et al. Risk factors for sporadic cryptosporidiosis among immunocompetent persons in the United States from 1999 to 2001. J Clin Microbiol 2004;42:2944--51. CDC. Diagnostic procedures for stool specimens. Atlanta, GA: US Department of Health and Human Services, CDC; 2007. Available at http://www.dpd.cdc.gov/dpdx/HTML/DiagnosticProcedures.htm. CyclosporiasisHerwaldt BL. The ongoing saga of U.S. outbreaks of cyclosporiasis associated with imported fresh produce: what Cyclospora cayetanensis has taught us and what we have yet to learn. In: Institute of Medicine. Addressing foodborne threats to health: policies, practices, and global coordination. Washington, DC: The National Academies Press; 2006:85--115, 133--40. Available at http://newton.nap.edu/catalog/11745.html#toc. Herwaldt BL. Cyclospora cayetanensis: a review, focusing on the outbreaks of cyclosporiasis in the 1990s. Clin Infect Dis 2000;31:1040--57. DiphtheriaDewinter LM, Bernard KA, Romney MG. Human clinical isolates of Corynebacterium diphtheriae and Corynebacterium ulcerans collected in Canada from 1999 to 2003 but not fitting reporting criteria for cases of diphtheria. Clin Microbiol 2005;43:3447--9. Domestic Arboviral Diseases, Neuroinvasive and NonneuroinvasiveCDC. Detection of West Nile Virus in Blood Donations---Puerto Rico, 2007. MMWR 2008;57:577--80. CDC. West Nile virus activity---United States, 2007. MMWR 2008;57:720--3. Enterohemorrhagic Escherichia coliBender JB, Hedberg CW, Besser JM, et al. Surveillance for Escherichia coli O157:H7 infections in Minnesota by molecular subtyping. N Engl J Med 1997;337:388--94. Brooks JT, Sowers EG, Wells JB, et al. Non-O157 Shiga toxin-producing Escherichia coli infections in the United States, 1983--2002. J Infect Dis 2005;192:1422--9. Crump JA, Sulka AC, Langer AJ, et al. An outbreak of Escherichia coli O157:H7 among visitors to a dairy farm. N Engl J Med 2002;347:555--60. Griffin PM, Mead PS, Sivapalasingam S. Escherichia coli O157:H7 and other enterohemorrhagic E. coli. In: Blaser MJ, Smith PD, Ravdin JI, Greenberg HB, Guerrant RL, eds. Infections of the gastrointestinal tract. Philadelphia, PA: Lippincott Williams & Wilkins; 2002:627--42. Mead PS, Griffin PM. Escherichia coli O157:H7. Lancet 1998;352:1207--12. Ehrlichiosis (Human Ehrlichioses as a result of infection with Ehrlichia and Anaplasma)Walker D. Rickettsiae and rickettsial infections: the current state of knowledge. CID 2007:45 (Suppl 1):539--44. Dumler JS, Madigan JE, Pusterla N, Bakken JS. Ehrlichioses in humans: epidemiology, clinical presentation, diagnosis, and treatment. CID 2007:45 (Suppl 1) 545--51. Demma LJ, Holman RC, McQuiston JH, Krebs JW, Swerdlow DL. Epidemiology of human ehrlichiosis and anaplasmosis in the United States, 2001--2002. Am J Trop Med Hyg 2005;73:400--9. Paddock CD, Childs JE. Ehrlichia chaffeensis: a prototypical emerging pathogen [Review]. J Clin Microbiol 2003;16:37--64. IJdo JW, Meek JI, Cartter ML, et al. The emergence of another tick-borne infection in the 12-town area around Lyme, Connecticut: human granulocytic ehrlichiosis. J Infect Dis 2000;181:1388--93. GiardiasisStuart JM, Orr HJ, Warburton FG, et al. Risk factors for sporadic giardiasis: a case-control study in southwestern England. Emerg Infect Dis 2003;9:229--33. CDC. Diagnostic procedures for stool specimens. Atlanta, GA: US Department of Health and Human Services, CDC; 2007. Available at http://www.dpd.cdc.gov/dpdx/HTML/DiagnosticProcedures.htm. GonorrheaCDC. Sexually transmitted diseases treatment guidelines, 2006. MMWR 2006;55(No. RR-11). CDC. Sexually transmitted diseases surveillance 2007 supplement: Gonococcal Isolate Surveillance Project (GISP) annual report 2007. Atlanta, GA: US Department of Health and Human Services, CDC; 2009. Available at http://www.cdc.gov/std/gisp2007. Datta SD, Sternberg M, Johnson RE, et al. Gonorrhea and chlamydia in the United States among persons 14 to 39 years of age, 1999 to 2002. Ann Int Med 2007;147:89--96. Haemophilus influenzae, Invasive DiseaseFry AM, Lurie P, Gidley M, Schmink S, Lingappa J, Rosenstein NE. Haemophilus influenzae type b (Hib) disease among Amish children in Pennsylvania: reasons for persistent disease. Pediatrics 2001;108:1--6. Hansen Disease (Leprosy)Britton WJ, Lockwood NJ. Leprosy. Lancet 2004;363:1209--19. Bruce S, Schroeder TL, Ellner K, Rubin H, Williams T, Wolf JE Jr. Armadillo exposure and Hansen’s disease: an epidemiologic survey in southern Texas. J Am Acad Dermatol 2000;43(2 Pt1):223--8. Hartzell JD, Zapor M, Peng S, Straight T. Leprosy: a case series and review. South Med J 2004;97:1252--6. Hastings R, Ed. Leprosy. 2nd ed. New York, NY: Churchill Livingstone; 1994. Joyce MP, Scollard DM. Leprosy (Hansen’s disease). In: Rakel RE, Bope ET, eds. Conn’s current therapy 2004: latest approved methods of treatment for the practicing physician. 56th ed. Philadelphia, PA: Saunders; 2004:100--5. Ooi WW, Moschella SL. Update on leprosy in immigrants in the United States: status in the year 2000. Clin Infect Dis 2001;32:930--7. Scollard DM, Adams LB, Gillis TP, Krahenbuhl JL, Truman RW, Williams DL. The continuing challenges of leprosy. Clin Microbio Rev, 2006;19: 338--81. Hantavirus Pulmonary SyndromeCDC. Hantavirus pulmonary syndrome---five states, 2006. MMWR 2006;55:627--9. Hemolytic Uremic Syndrome, PostdiarrhealBanatvala N, Griffin PM, Greene KED, et al. The United States Prospective Hemolytic Uremic Syndrome Study; microbiologic, serologic, clinical, and epidemiologic findings. J Infect Dis 2001;183:1063--70. Mahon BE, Griffin PM, Mead PS, Tauxe RV. Hemolytic uremic syndrome surveillance to monitor trends in infection with Escherichia coli O157:H7 and other Shiga toxin-producing E. coli [Letter]. Emerg Infect Dis 1997;3:409--12. Hepatitis AArmstrong GL, Bell BP. Hepatitis A virus infections in the United States: model-based estimates and implications for childhood immunization. Pediatrics 2002;109:839--45. Bell BP, Kruszon-Moran D, Shapiro CN, Lambert SB, McQuillan GM, Margolis HS. Hepatitis A virus infection in the United States: serologic results from the Third National Health and Nutrition Examination Survey. Vaccine 2005;23:5798--806. Wasley A, Samandari T, Bell BP. Incidence of hepatitis A in the United States in the era of vaccination. JAMA 2005;294:194--201. Wasley A, Fiore A, Bell BP. Hepatitis A in the era of vaccination. Epidemiol Rev 2006;28:101--11. Hepatitis BArmstrong GL, Mast EE, Wojczynski M, Margolis HS. Childhood hepatitis B virus infections in the United States before hepatitis B immunization. Pediatrics 2001;108:1123--8. Shepard CW, Simard EP, Finelli L, Fiore A, Bell BP. Hepatitis B virus infection: epidemiology and vaccination. Epidemiol Rev 2006;28:112--25. Goldstein ST, Alter MJ, Williams IT, et al. Incidence and risk factors for acute hepatitis B in the United States, 1982--1998: implications for vaccination programs. J Infect Dis 2002;185:713--9. McQuillan GM, Coleman PJ, Kruszon-Moran D, Moyer LA, Lambert SB, Margolis HS. Prevalence of hepatitis B virus infection in the United States: The National Health and Nutrition Examination Surveys, 1976 through 1994. Am J Public Health 1999;89:14--8. Thompson NA, Perez JF, Moorman AC, Holmberg SD. Nonhospital health-care--associated hepatitis B and C transmission --- United States, 1998--2008. Ann Intern Med 2009;150:33--9. Hepatitis CArmstrong GL, Wasley A, Simard EP, McQuillan GM, Kuhnert WL, Alter MJ. The prevalence of hepatitis C virus infection in the United States, 1999 through 2002. Ann Intern Med 2006;144):705--14. Armstrong GA, Alter MJ, McQuillan GM, Margolis HS. The past incidence of hepatitis C virus infection: implications for the future burden of chronic liver disease in the United States. Hepatology 2000;31:777--82. Shepard CW, Finelli L, Alter MJ. The global epidemiology of hepatitis C. Lancet Infect Dis 2005;5:558--67. Thompson NA, Perez JF, Moorman AC, Holmberg SD. Nonhospital health-care associated hepatitis B and C transmission ---United States, 1998--2008. Ann Intern Med. 2009;150:33--9. Influenza-Associated Pediatric MortalityBhat N, Wright JG, Broder KR, et al. Influenza-associated deaths among children in the United States, 2003--2004. N Engl J Med 2005;352:2559--67. Council of State and Territorial Epidemiologists. Influenza-associated pediatric mortality, 2004. Atlanta, GA: Council of State and Territorial Epidemiologists; 2004. Available at http://www.cste.org/PositionStatementsResolutions2.htm. Council of State and Territorial Epidemiologists. Position statement 04-ID-04: influenza-associated pediatric mortality 2004. Atlanta, GA: Council of State and Territorial Epidemiologists; 2004. Available at http://www.cste.org/ps/2004pdf/04-ID-04-final.pdf. Guarner J, Paddock CD, Shieh WJ, et al. Histopathologic and immunohistochemical features of fatal influenza virus infection in children during the 2003--2004 season. Clin Infect Dis 2006:43;132--40. LegionellosisFields BS, Benson RF, Besser RE. Legionella and Legionnaires’ disease: 25 years of investigation. Clin Microbiol Rev 2002;15:506--26. European Working Group on Legionella Infections. European guidelines for control and prevention of travel associated Legionnaires’ disease. London, UK: United Kingdom Health Protection Agency; 2005. Joseph CA. Legionnaires’ disease in Europe 2000--2002. Epidemiol Infect 2004;132:417--24. Marston BJ, Lipman HB, Breiman RF. Surveillance for Legionnaires’ disease: risk factors for morbidity and mortality. Arch Intern Med 1994;154:2417--22. Neil K, Berkelman R. Increasing incidence of legionellosis in the United States: changing epidemiological trends. Clin Infect Dis 2008;47:591--9. ListeriosisGottlieb SL, Newbern EC, Griffin PM, et al. Multistate outbreak of listeriosis linked to turkey deli meat and subsequent changes in US regulatory policy. Clin Infect Dis 2006;42:29--36. Mead PS, Dunne EF, Graves L, et al. Nationwide outbreak of listeriosis due to contaminated meat. Epidemiol Infect 2006;134:744--51. Mead PS, Slutsker L, Dietz V, et al. Food-related illness and death in the United States. Emerg Infect Dis 1998;5:607--25. Slutsker L, Schuchat A. Listeriosis in humans. In: Ryser ET Marth EH, eds. Listeria, listeriosis, and food safety. 2nd ed. New York, NY: Marcel Dekker, Inc.; Little, Brown and Company; 1999:75--95. Voetsch AC, Angulo FJ, Jones TF, et al. Reduction in the incidence of invasive listeriosis in Foodborne Diseases Active Surveillance Network Sites, 1996--2003. Clin Infect Dis 2007;44:513--20. Lyme DiseaseStafford KC III. Tick management handbook: an integrated guide for homeowners, pest control operators, and public health officials for the prevention of tick-associated disease. New Haven, CT: Connecticut Agricultural Experiment Station; 2004. Available at http://www.cdc.gov/ncidod/dvbid/lyme/resources/handbook.pdf. Hayes EB, Piesman J. How can we prevent Lyme disease? N Engl J Med 2003;348:2424--30. Wormser GP, Dattwyler RJ, Shapiro ED, et al. The clinical assessment, treatment, and prevention of Lyme disease, human granulocytic anaplasmosis, and babesiosis: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis 2006;43:1089--1134. Medical Letter. Treatment of Lyme disease. Med Lett Drugs Ther 2005;47:41--3. CDC. Caution regarding testing for Lyme disease. MMWR 2005;54:125. MalariaBaird JK. Effectiveness of antimalarial drugs. N Engl J Med 2005;352:1565--77. Chen LH, Keystone JS. New strategies for the prevention of malaria in travelers. Infect Dis Clin N Amer 2005;19:185--210. Guinovart C, Navia MM, Tanner M, et al. Malaria: burden of disease. Curr Mol Med 2006;6:137--40. Leder K, Black J, O’Brien D, et al. Malaria in travelers: a review of the GeoSentinel Surveillance Network. Clin Infect Dis 2004;39:1104--12. MeaslesPapania M, Hinman A, Katz S, Orenstein W, McCauley M, eds. Progress toward measles elimination---absence of measles as an endemic disease in the United States. J Infect Dis 2004;189(Suppl 1):S1--257. Rota PA, Liffick SL, Rota JS, et al. Molecular epidemiology of measles viruses in the United States, 1997--2001. Emerg Infect Dis 2002;8:902--8. De Serres G, Gay NJ, Farrington CP. Epidemiology of transmissible diseases after elimination. Am J Epidemiol 2000;151:1039--48. Meningococcal DiseaseRosenstein NE, Perkins BA, Stephens DS, et al. Meningococcal disease. N Engl J Med 2001;344:1378--88. Rosenstein NE, Perkins BA, Stephens DS, et al. The changing epidemiology of meningococcal disease in the United States, 1992--1996. J Infect Dis 1999;180:1894--901. MumpsCDC. Mumps epidemic---Iowa, 2006. MMWR 2006;55:366--8. CDC. Update: mumps activity---United States, January 1--October 7, 2006. MMWR 2006;55:1152--3. Harling R, White JM, Ramsay ME, et al. The effectiveness of the mumps component of the MMR vaccine: a case control study. Vaccine 2005;23:4070--4. Schaffzin JK, Pollock L, Schulte C, et al. Effectiveness of previous mumps vaccination during a summer camp outbreak. Pediatrics 2007;120:e862--8. Dayan G, Quinlisk P, et al. Recent resurgence of mumps in the United States. New Engl J Med 2008;358:1580--9. Novel influenza A virusOlsen CW. The emergence of novel swine influenza viruses in North America. Virus Res 2002;85:199--210. Dowdle, WR. Influenza pandemic periodicity, virus recycling, and the art of risk assessment. Emerg Infect Dis. 2006;12:34--9. National Pork Board. Influenza: pigs, people and public health. Des Moines, IA: National Pork Board; 2004. Available at: http://www.porkboard.org/PorkScience/Documents/PUBLICHEALTH%20influenza.pdf. PertussisBisgard KM, Rhodes P, Connelly BL, et al. Pertussis vaccine effectiveness among children 6 to 59 months of age in the United States, 1998--2001. Pediatrics 2005;116:e285--94. Bisgard KM, Pascual FB, Ehresmann KR, et al. Infant pertussis: who was the source? Pediatr Infect Dis J 2004;23:985--9. CDC. Pertussis---United States, 2001--2003. MMWR 2005;54:1283--6. Lee GM, Lebaron C, Murphy TV, Lett S, Schauer S, Lieu TA. Pertussis in adolescents and adults: should we vaccinate? Pediatrics 2005;115:1675--84. PlagueCDC. Imported plague---New York City, 2002. MMWR 2003;53:725--8. Enscore RE, Biggerstaff BJ, Brown TL, et al. Modeling relationships between climate and the frequency of human plague cases in the southwestern United States, 1960--1997. Am J Trop Med Hyg 2002;66:186--96. Inglesby TV, Dennis DT, Henderson DA, et al. Plague as a biological weapon: medical and public health management. Working Group on Civilian Biodefense. JAMA 2000;283:2281--90. Dennis DT, Gage KL, Gratz N, Poland JD, Tikhomirov E. Plague manual: epidemiology, distribution, surveillance and control. Geneva, Switzerland: World Health Organization; 1999. PoliomyelitisAlexander LN, Seward JF, Santibanez TA, et al. Vaccine policy changes and epidemiology of polio in the United States. JAMA 2004;292:1696--702. Q FeverTissot-Dupont D, Raoult D: Q Fever. Infect Dis Clin North Am 2008;22:505--14. Parker N, Barralet J, Bell A. Q fever. The Lancet 2006 367;511:679--88. McQuiston JH, Holman RC, McCall CL, Childs JE, Swerdlow DL, Thompson HA. National surveillance and the epidemiology of Q fever in the United States, 1978--2004. Am J Trop Med Hyg 2006;75:36--40. Mcquiston JH, Nargund VN, Miller JD, Priestly R, Shaw EI, Thompson HA. Prevalence of antibodies to Coxiella burnetii among veterinary school dairy herds in the United States, 2003. Vector Borne Zoonotic Dis 2005;90--1. Raoult D, Tissot-Dupont H, Foucault C, et al. Q fever 1985--1998. Clinical and epidemiologic features of 1,383 infections [Review]. Medicine 2000:79:109--25. Bernard KW, Parham GL, Winkler WG, Helmick CG. Q fever control measures: recommendations for research facilities using sheep. Infection Control 1982;3:461--65. Rabies, Animal and HumanBlanton JD, Hanlon CA, Ruprrecht CE. Rabies surveillance in the United States during 2006. J Am Vet Med Assoc 2007;231:540--56. Rocky Mountain Spotted FeverWalker D. Rickettsiae and rickettsial infections: the current state of knowledge. Clin Infect Dis 2007:45 (Suppl 1):539--44. Chapman AS, Murphy SM, Demma LJ, et al. Rocky Mountain spotted fever in the United States, 1997--2002. Vector-borne Zoonotic Dis. 2006;6:170--8. Demma LJ, Traeger MS, Nicholson WL, et al. Rocky Mountain spotted fever from an unexpected tick reservoir in Arizona. N Engl J Med 2005;353:587--94. Thorner AR, Walker DH, Petri WA. Rocky Mountain spotted fever [Review]. Clin Infect Dis 1998;27:1353--60.. RubellaReef S, Cochi S, eds. The evidence for the elimination of rubella and congenital rubella syndrome in the United States: a public health achievement. Clin Infect Dis 2006;43(Suppl 3):S123--68. SalmonellosisBraden CR. Salmonella enterica serotype Enteritidis and eggs: a national epidemic in the United States. Clin Infect Dis 2006;43:512--7. Olsen SJ, Bishop R, Brenner FW, et al. The changing epidemiology of Salmonella: trends in serotypes isolated from humans in the United States, 1987--1997. J Infect Dis 2001;183:756--61. Voetsch AC, Van Gilder TJ, Angulo FJ, et al. FoodNet estimate of burden of illness caused by nontyphoidal Salmonella infections in the United States. Clin Infect Dis 2004;38(Suppl 3):S127--34. ShigellosisShane A, Crump J, Tucker N, Painter J, Mintz E. Sharing Shigella: risk factors and costs of a multi-community outbreak of shigellosis. Arch Pediatr Adolesc Med 2003;157:601--3. Gupta A, Polyak CS, Bishop RD, Sobel J, Mintz ED. Laboratory-confirmed shigellosis in the United States, 1989--2002: epidemiologic trends and patterns. Clin Infect Dis 2004;38:1372--7. Sivapalasingam S, Nelson JM, Joyce K, Hoekstra M, Angulo FJ, Mintz ED. A high prevalence of antimicrobial resistance among Shigella isolates in the United States, 1999--2002. Antimicrob Agents Chemother 2006;50:49--54. SmallpoxLi Y, Carroll DS, Gardner SN, Walsh MC, Vitalis EA, Damon IK. On the origin of smallpox: correlating variola phylogenics with historical smallpox records. Proc Natl Acad Sci U S A. 2007 Oct 2;104:15787--92. Streptococcal Disease, Invasive, Group AO’Loughlin RE, Roberson A, Cieslak PR, et al. The epidemiology of invasive group A streptococcal infections and potential vaccine implications, United States, 2000--2004. Clin Infect Dis 2007; 45:853--62. CDC. Active Bacterial Core Surveillance report. Emerging Infections Program Network. Group A streptococcus, 2007-Provisional. Atlanta, GA: US Department of Health and Human Services, CDC; 2008. Available at http://www.cdc.gov/ncidod/dbmd/abcs/survreports/GAS_2007_provisional.pdf. Carapetis JR. Steer AC. Mulholland EK. Weber M. The global burden of group A streptococcal diseases. Lancet Infect Dis 2005;5:685--94. Jordan HT, Richards CL, Burton DC, Thigpen MC, Van Beneden CA. Group A streptococcal disease in long-term care facilities: descriptive epidemiology and potential control measures. Clin Infect Dis 2007;45:742--52. CDC. Investigating clusters of group A streptococcal disease. Atlanta, GA: US Department of Health and Human Services, CDC; 2005. Available at http://www.cdc.gov/strepAcalculator. The Prevention of Invasive Group A Streptococcal Infections Workshop Participants. Prevention of invasive group A streptococcal disease among household contacts of case patients and among postpartum and postsurgical patients: recommendations from the Centers for Disease Control and Prevention. Clin Infect Dis 2002;35:950--9. Streptococcal Toxic-Shock SyndromeBisno AL. Brito MO. Collins CM. Molecular basis of group A streptococcal virulence. Lancet Infect Dis 2003;3:191--200. O’Loughlin RE, Roberson A, Cieslak PR, et al. The epidemiology of invasive group A streptococcal infections and potential vaccine implications, United States, 2000-2004. Clin Infect Dis 2007; 45:853--62. Stevens DL. Streptococcal toxic shock syndrome associated with necrotizing fasciitis. Annu Rev Med 2000;51:271--88. The Prevention of Invasive Group A Streptococcal Infections Workshop Participants. Prevention of invasive group A streptococcal disease among household contacts of case patients and among postpartum and postsurgical patients: recommendations from the Centers for Disease Control and Prevention. Clin Infect Dis 2002;35:950--9. Streptococcus pneumoniae, Invasive, Drug-ResistantClinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing: 15th informational supplement [No. M100-S15]. Wayne, PA: National Committee for Clinical Laboratory Standards; 2005. Flannery B, Schrag S, Bennett NM, et al. Impact of childhood vaccination on racial disparities in invasive Streptococcus pneumoniae infections. JAMA 2004;291:2197--203. Kyaw MH, Lynfield R, Schaffner W, et al. Effect of introduction of the pneumococcal conjugate vaccine on drug-resistant Streptococcus pneumoniae. N Engl J Med 2006;354:1455--63. Poehling KA, Talbot TR, Griffin MR, et al. Invasive pneumococcal disease among infants before and after introduction of pneumococcal conjugate vaccine. JAMA 2006;295:1668--74. Ray GT, Whitney CG, Fireman BH, Ciuryla V, Black SB. Cost-effectiveness of pneumococcal conjugate vaccine: evidence from the first 5 years of use in the United States incorporating herd effects. Pediatr Infect Dis J 2006;25:494--501. Syphilis, CongenitalCDC. Congenital syphilis---United States, 2002. MMWR 2004;53:716--9. Syphilis, Primary and SecondaryCDC. The National Plan to Eliminate Syphilis from the United States. Atlanta, GA: US Department of Health and Human Services, CDC; 1999. CDC. The National Plan to Eliminate Syphilis from the United States. Atlanta, GA: US Department of Health and Human Services, CDC; 2006. CDC. Primary and secondary syphilis---United States, 2003--2004. MMWR 2006;55:269--73. CDC. Sexually transmitted disease surveillance, 2007. Atlanta, GA: US Department of Health and Human Services, CDC; 2008. Available at http://www.cdc.gov/std/stats. Heffelfinger JD, Swint EB, Berman SB, Weinstock HS. Trends in primary and secondary syphilis among men who have sex with men in the United States. Am J Public Health 2007;97:1076--83. TetanusCDC. Tetanus---Puerto Rico, 2002. MMWR 2002;51:613--5. McQuillan GM, Kruszon-Moran D, Deforest A, Chu SY, Wharton M. Serologic immunity to diphtheria and tetanus in the United States. Ann Intern Med 2002;136:660--6. TrichinellosisCDC. Trichinellosis associated with bear meat---New York and Tennessee, 2003. MMWR 2004;53:606--10. Moorhead A, Grunenwald PE, Dietz VJ, Schantz PM. Trichinellosis in the United States, 1991--1996: declining but not gone. Am J Trop Med Hyg 1999;60:66--9. Burke R, Masuoka P, Darwin Murrell K. Swine Trichinella infection and geographic information system tools. Emerg Infect Dis 2008;14:1109--11. Gamble HR, Bessonov AS, Cuperlovic K, et al. International Commission on Trichinellosis: recommendations on methods for the control of Trichinella in domestic and wild animals intended for human consumption. Vet Parasitol 2000;93:393--408. TuberculosisCDC. Reported tuberculosis in the United States, 2003. Atlanta, GA: US Department of Health and Human Services, CDC; 2004. Available at http://www.cdc.gov/nchstp/tb. CDC. Trends in tuberculosis---United States, 2004. MMWR 2005;54:245--9. Saraiya M, Cookson ST, Tribble P, et al. Tuberculosis screening among foreign-born persons applying for permanent US residence. Am J Public Health 2002;92:826--9. Talbot EA, Moore M, McCray E, Binkin NJ. Tuberculosis among foreign-born persons in the United States, 1993--1998. JAMA 2000;284:2894--900. TularemiaCDC. Outbreak of tularemia among commercially distributed prairie dogs, 2002. MMWR 2002;51:688, 699. CDC. Tularemia---United States, 1990--2000. MMWR 2002;51:182--4. Dennis DT, Inglesby TV, Henderson DA, et al. Tularemia as a biological weapon: medical and public health management. JAMA 2001;285:2763--73. Feldman KA, Enscore RE, Lathrop SL, et al. Outbreak of primary pneumonic tularemia on Martha’s Vineyard. N Engl J Med 2001;345:1219--26. Petersen JM, Schriefer ME. Tularemia: emergence/re-emergence. Vet Res 2005;36:455--67. Typhoid FeverCrump JA, Kretsinger K, Gay K, et al. Clinical response and outcome of infection with Salmonella enterica serotype Typhi with decreased susceptibility to fluoroquinolones: a United States FoodNet multicenter retrospective cohort study. Antimicrob Agents Chemother 2008;52:1278--84. Gupta S, Medalla F, Omondi MW, et al. Laboratory-based surveillance for paratyphoid fever in the United States: travel and antimicrobial resistance. Clin Infect Dis; 2008;46:1656-63. Kubota K, Barrett TJ, Hunter S et al. Analysis of Salmonella serotype Typhi pulsed-field gel electrophoresis patterns associated with international travel. J Clin Micro 2005;43:1205--9. Olsen SJ, Bleasdale SC, Magnano AR, et al. Outbreaks of typhoid fever in the United States, 1960--1999. Epidemiol Infect 2003;130:13--21. Steinberg EB, Bishop RB, Dempsey AF, et al. Typhoid fever in travelers: who should be targeted for prevention? Clin Infect Dis 2004;39:186--91. VaricellaCDC. Public health response to varicella outbreaks---United States, 2003--2004. MMWR 2006;55:993--5. CDC. Varicella surveillance practices---United States, 2004. MMWR 2006;55:1126--9. Seward JF, Zhang JX, Maupin TJ, Mascola L, Jumaan AO. Contagiousness of varicella in vaccinated cases: a household contact study. JAMA 2004;292:704--8. Vancomycin-Intermediate Staphylococcus aureus Infection (VISA)/Vancomycin-Resistant Staphylococcus aureus Infection (VRSA)Fridkin SK, Hageman J, McDougal LK, et al. Vancomycin-Intermediate Staphylococcus aureus Epidemiology Study Group. Epidemiological and microbiological characterization of infections caused by Staphylococcus aureus with reduced susceptibility to vancomycin, United States, 1997--2001. Clin Infect Dis 2003;36:429--39. Chang S, Sievert DM, Hageman JC, et al. Vancomycin-Resistant Staphylococcus aureus Investigative Team. Infection with vancomycin-resistant Staphylococcus aureus containing the vanA resistance gene. N Engl J Med 2003;348:1342--7. Whitener CJ, Park SY, Browne FA, et al. Vancomycin-resistant Staphylococcus aureus in the absence of vancomycin exposure. Clin Infect Dis 2004;38:1049--55. Weigel LM, Clewell DB, Gill SR, et al. Genetic analysis of a high-level vancomycin-resistant isolate of Staphylococcus aureus. Science 2003;302:1569--71. McDonald LC, Hageman JC. Vancomycin intermediate and resistant Staphylococcus aureus: what the nephrologist needs to know. Nephrol News Issues 2004;8:63--4, 66--7, 71--2. VibriosisDaniels NA, MacKinnon L, Bishop R, et al. Vibrio parahaemolyticus infections in the United States, 1973-1998. J Infect Dis 2000;181:1661--6. Dechet A, Yu PA, Koram N, Painter J. Nonfoodborne vibrio infections: an important cause of morbidity and mortality in the United States, 1997--2006 McLaughlin JB, DePaola A, Bopp CA, et al. Outbreak of Vibrio parahaemolyticus gastroenteritis associated with Alaskan oysters. N Engl J Med 2005;353:1463--70. Shapiro RL, Altekruse S, Hutwagner L, et al. The role of Gulf Coast oysters in warmer months in Vibrio vulnificus infections in the United States, 1998--1996. J Infect Dis 1998;178:752--9. Date last reviewed: 5/29/2009 Syndicated Content Details:
Source URL: http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5653a1.htm Source Agency: Centers for Disease Control and Prevention (CDC) Captured Date: 2016-05-23 22:08:29.0
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||