
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Appendix F: Management of the IUD when a Cu-IUD or an LNG-IUD User Is Found To Have Pelvic Inflammatory Disease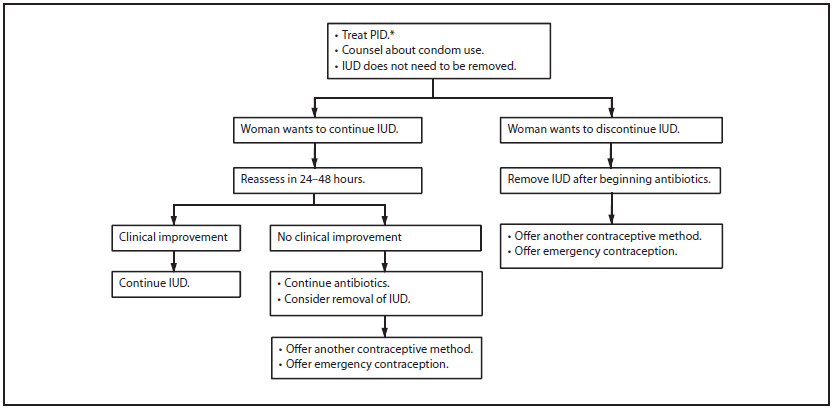
Abbreviations: Cu-IUD = copper-containing IUD; IUD = intrauterine device; LNG-IUD = levonorgestrel-releasing IUD; PID = pelvic inflammatory disease. * Treat according to CDC's STD Treatment Guidelines (available at http://www.cdc.gov/std/treatment). Alternate Text: Appendix F shows a flow chart describing the management of an intrauterine device when a woman using a copper-containing IUD or a levonorgestrel-releasing IUD is found to have pelvic inflammatory disease. Syndicated Content Details:
Source URL: http://www.cdc.gov/mmwr/preview/mmwrhtml/rr6205a7.htm Source Agency: Centers for Disease Control and Prevention (CDC) Captured Date: 2016-05-23 22:24:52.0
|
©2017 ARCHES
Technology. All Rights Reserved.